Belgian Celyad granted an exclusive license to Osaka based ONO Pharmaceutical for the development and commercialisation of Celyad’s unique allogeneic cell therapy in Japan, Korea and Taiwan.
 This license agreement opens new markets to Celyad and its NKR-2 T-cell cancer immunotherapy. In addition to a deal worth up to €282M, Celyad will also receive double digit royalties based on net sales of the licensed product in Japan-based ONO’s territories.
This license agreement opens new markets to Celyad and its NKR-2 T-cell cancer immunotherapy. In addition to a deal worth up to €282M, Celyad will also receive double digit royalties based on net sales of the licensed product in Japan-based ONO’s territories.
Celyad’s Natural Killer Receptor (NKR) based T-Cell platform targets a wide range of solid and hematological tumors, and is currently in Phase I trials for acute myeloid leukemia and multiple myeloma patients.
Unlike traditional CAR-T cell therapy, which target only one tumor antigen, NK cell receptors enable a single receptor to recognise multiple tumor antigens. The trial is designed to assess the safety and feasibility of NKR-2, with secondary endpoints including clinical activity.
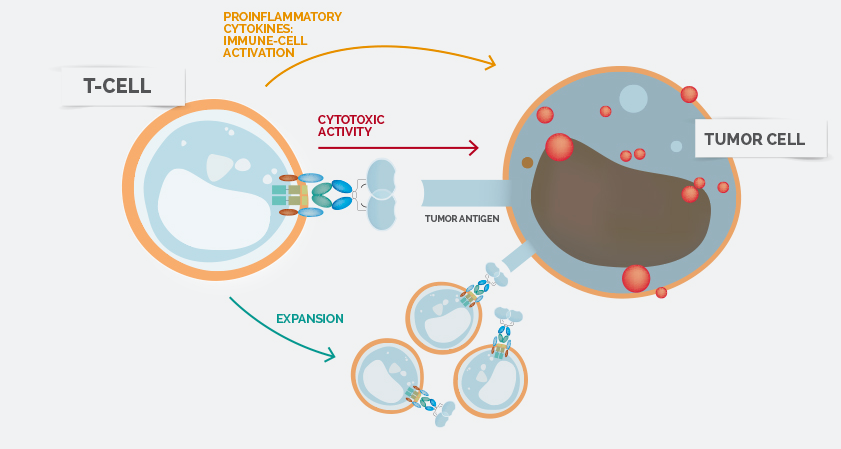
Additional benefits of this approach include the fact that the NKR-2 program does not employ patient pre-conditioning of the immune system, thereby avoiding the toxicities associated with chemotherapy and allowing the immune system to remain intact.
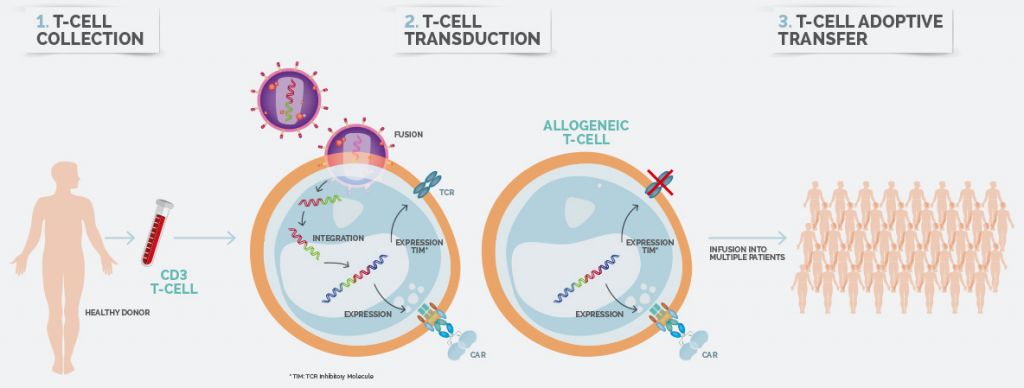
Celyad’s allogeneic platform engineers the T-Cells of healthy donors, that also express TCR Inhibitory Molecules (TIMs), to avoid having the engineered donor cells be rejected by the patient’s normal tissues.
There are significant cost-related benefits to an allogenic platform too, which is the basis for an ‘off-the shelf‘ therapy. As multiple treatments are be made from a single donor, the cost of each dose is lowered.
And whilst this Belgian biotech has had a disappointing quarter for its Cardiology program (as did a few biotechs), its CAR-T work has instead seen encouraging progress. Celyad also holds key patents in the CAR-T field, which was also one of the main reasons why it successfully IPOed on the NASDAQ and raised over $100M.
This is a difficult period for CAR-T, the field of which has faced a lot of hype and then just some last week received some upsetting news in the US.
However, this partnership is the next solid step for other CAR-T biotechs to ‘get back on the horse’, so to speak.
George Rawadi spoke to Labiotech at our conference Refresh in May…
Feature Image Credit: Celyad





