This article was originally published in July 2018 and has since been updated with the most recent developments in blood sugar testing.
Will the daily routine of finger pricking to monitor blood sugar levels finally come to an end for the millions living with diabetes?
Diabetes affects over 537 million adults worldwide. People with diabetes have to test their blood sugar levels several times a day, usually by pricking their finger with a lancet. This can be uncomfortable and painful for many, which can result in less frequent testing and consequently worse control of blood sugar levels.
The last decade has seen the rise of blood sugar monitors that are installed by pricking the skin and only need replacing every few weeks. One of the best sellers is FreeStyle Libre, developed by Abbott Diabetes Care in the U.S., which measures glucose levels in the interstitial fluid between the cells right under the skin.
Many companies around the world aim to make the lives of millions of people with diabetes easier by developing non-invasive methods of glucose monitoring. Making these methods as accurate as traditional test strips is a tough undertaking, however. One high-profile example is the Google Contact Lens, which was abandoned in 2018 because its method of reading glucose levels in tears wasn’t consistent enough.
Companies that can crack the challenges of measuring glucose accurately and affordably with no needles stand to reap a share of the fast-growing market of blood glucose monitors. Here is a shortlist of some of the most exciting candidates in the market and in the pipeline.
Table of contents
D-Base
D-Base is a shoebox-sized blood sugar monitor developed by the German firm DiaMonTech. The device measures blood sugar levels by beaming an infrared laser through the skin of a finger and causing glucose in the skin to convert the light to heat. The machine then calculates glucose levels based on the increase of heat in the skin. The increase in temperature is too minimal to be noticed by the user.
In 2019, D-Base was approved in the EU for use by medical professionals in clinical trials and diabetes centers. Earlier this year, the company attracted $5 million in funding — bringing its total to $20 million raised — to help bring D-Base to the U.S. market. Additionally, DiaMonTech is working on smaller versions of the technology, including a handheld device called D-Pocket as well as the small D-Sensor that can be used in wearable devices.

Eversense
Developed by U.S. company Senseonics and distributed by Ascensia Diabetes Care, Eversense is a subcutaneous implant that continuously monitors blood glucose levels. Although it initially needs to be installed under the skin by a doctor, the sensor can last for up to three months before needing a replacement.
Eversense measures glucose in the interstitial fluid under the skin of the upper arm by using a polymer that fluoresces in response to the levels of blood sugar. The data is then sent to a transmitter that displays the blood glucose levels in real time.
The device received U.S. Food and Drug Administration (FDA) approval in 2018 and the company struck a deal with Roche to distribute the sensor. A six-month version of the implant was approved in Europe in 2017 and in the U.S. in early 2022. Senseonics is also working on an implant that can last for up to one year.
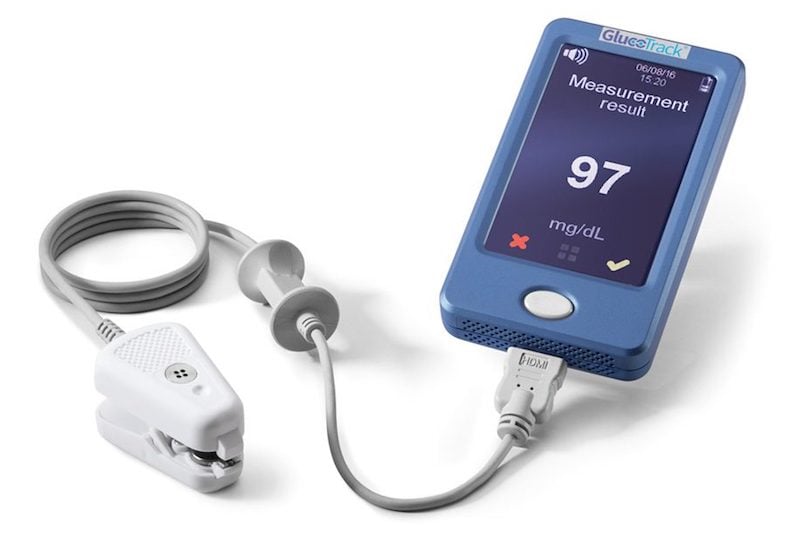
GlucoTrack
Developed by the U.S.–Israeli company Integrity Applications — now rebranded as GlucoTrack, Inc. — the GlucoTrack device can monitor blood sugar levels through a combination of ultrasonic, electromagnetic and thermal waves. To provide a readout, the sensor is clipped on the ear.
The device is indicated for adults with type 2 diabetes and is marketed in Europe. Integrity Applications now aims to expand into the U.S. and is developing the second generation of GlucoTrack, which consists of a wireless ear clip sensor paired with a smartphone. Initial study results of the Gen 2 monitor have shown good performance and accuracy.
glucoWISE
glucoWISE is a sensor under development that could measure blood glucose levels by just placing it on the skin between the thumb and forefinger. The real-time measurements are then sent directly to a smartphone app.
By using a specific frequency of radio waves to measure blood glucose levels, the developers believe the device would be more accurate than are other wireless glucose monitors.
The U.K. firm behind the technology, MediWise, was acquired by the Canadian smart materials and photonics specialist Meta Materials in 2018. Meta Materials is continuing the development of glucoWISE, which has already been tested in two small-scale human trials. Earlier this year, the device was granted a U.S. patent for non-invasive glucose sensing system.
NovioSense
NovioSense is a Dutch startup working on a blood sugar monitor that is placed under the lower eyelid, from where it can wirelessly send glucose measurements directly to a smartphone. The device consists of a flexible metal coil of just two centimeters in length with nanosensors contained inside. In turn, the coil is covered by a protective layer of soft hydrogel.
The coil can measure minute-to-minute changes in the glucose levels of tear fluid by using the same enzyme technology on which conventional glucose strip tests are based. According to results from a clinical study published in 2020, the device is comparable in accuracy to the FreeStyle Libre.

Occuity Indigo
As an eye-focused blood glucose meter, Occuity Indigo takes a literal approach to the expression “the eyes are windows to the soul.” Rather than measuring tear fluid, the U.K. developer Occuity looks within the eyeball — similarly to the Google Contact Lens — as it is a transparent, stable environment whose glucose levels correlate with those of the blood.
The Occuity Indigo sends a faint beam of light into the eyeball and measures the light that bounces back into the device. It can infer glucose levels in the eye based on the refraction of the returning light.
The technology, which is still in research and development, was crowdfunded on Seedrs. Occuity is also developing a similar device that can screen people at risk of developing diabetes and other health conditions in the future.

SugarBEAT
SugarBEAT, developed by U.K. biotech Nemaura Medical, is a replaceable skin patch attached to a transmitter suitable for people with both type 1 and type 2 diabetes as well as pre-diabetes. It measures blood glucose levels non-invasively by passing a low-level electric current across the skin that draws out a sample of the interstitial fluid, found just below the skin.
The transmitter, which is rechargeable, sends data to the user’s phone every five minutes using Bluetooth and the readings can be monitored using an accompanying app.
Nemaura’s smart device already has marketing approval in the EU, and the company has submitted a Premarket Approval to the U.S. FDA, which is currently in review.






