Newsletter Signup - Under Article / In Page
"*" indicates required fields
Everything you need to know about what checkpoint inhibitors are and why they have caused a wave of hype for cancer immunotherapies.
Since the approval of the first PD-1 checkpoint inhibitor, Keytruda, in 2014, there has been an explosion of the numbers of trials testing these drugs, both alone and in combination. The latest figure, coming from the Cancer Research Institute (CRI) in the US, sets the number in over 1,500 trials.
Checkpoint inhibitors have yielded impressive results in a certain percentage of patients with cancer. Especially PD-1 and PD-L1 checkpoint inhibitors, which are now in the pipelines o all major big pharma. This has fueled the expectations on immunotherapies that activate and direct the immune system against cancer cells.
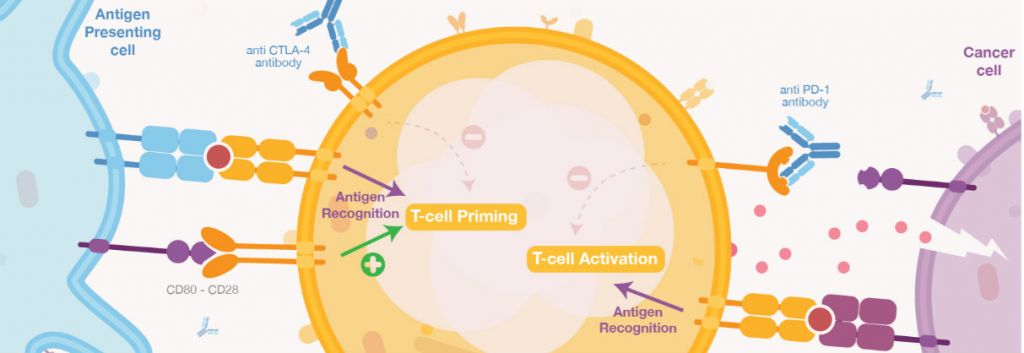
But given most of these approaches are still in early stages of research, it is important to not let hype drive expectations too high and take the time to understand exactly how these drugs work, with their benefits as well as their side effects and other disadvantages like expensive prices. There is a lot going on in the field, so we decided to put all the essentials in this infographic to make it easier to process for everyone. Enjoy!

What do you think? Are you hyped or skeptic about checkpoint inhibitors? Let us know in the comments below!

Etienne Raimondeau just completed his PhD in Biochemistry & Structural Biology at EMBL. When not pipetting, he enjoys communicating science through illustrations! You can find more of his work here.
Oncology R&D trends and breakthrough innovations