Arthur Lahr, CEO of Kiadis Pharma, explains the promising cell therapy the company is developing and discusses how the dutch biotech ecosystem has evolved.
Arthur Lahr has been part of the Dutch biotech scene for over 15 years now. The former Chief Strategy Officer of Crucell, which was sold to J&J in 2011, is now leading Kiadis Pharma as it enters Phase III and approaches the market. “I joined in January and took over as CEO in April, as part of the transition plan,” he explains.
Kiadis Pharma is developing a T-cell immunotherapy that focuses on improving the only therapy that can currently cure some of the patients with blood cancer, hematopoietic stem cell transplantation (HSCT). “We make those transplants safer for those patients,” Lahr says. With the inflated expectations for immuno-oncology being exposed to big challenges, especially regarding severe side-effects, Kiadis Pharma might have found a niche in developing products to make HSCT treatments safer for patients.
“If we focus on blood cancers, there are of course many therapies in development to put patients in remission, to get the tumor to disappear,” says Lahr. “But so far, there is no magic bullet that really cures patients long term. After a certain period of time, most patients relapse. CAR-T, of course, has huge potential to put more patients in remission, but I have to say that it has yet to be seen whether there will be a cure.”
Read on to learn how this technology works and how it could disrupt the market.
How does the technology of Kiadis Pharma work?
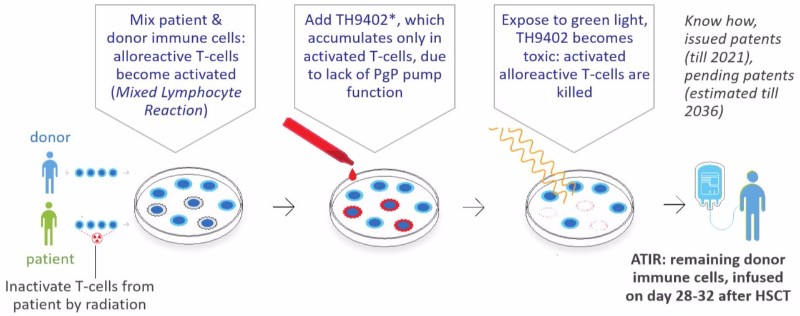
What we have is a patient-specific cell therapy that is given as an adjunctive treatment to a hematopoietic stem cell transplant. It will take 6-12 months for the stem cells in the transplants to regrow into a full immune system. And during those 6-12 months, patients are exposed. Our T-cell product, ATIR, can bridge that period and give immediate protection against relapse and infection without the need for a prophylactic immunosuppressant.
The problem with giving T-cells, though, is that they can cause Graft-versus-Host-Disease. Our products have been depleted of those T-cells that can cause GvHD. The protocol consists of giving the patient CD34+ stem cells, which is a “safe” HSCT transplant. And then a month later, we give them ATIR, which just contains T-cells that provide protection but not T-cells that can attack the patient.
Given all the possible applications, why are you going after leukemia and thalassemia?
Transplantation is often the only last resort available to patients, but the safety of bone marrow transplants in blood cancers is a huge issue. The best scenario would be for patients to find a matched donor, but that’s not available to about a third of patients. There are currently around 28,000 patients in the US and Europe that could benefit from an approach like ours, and leukemia is the biggest portion of that. It’s roughly 70%.
Our study was done in patients with acute leukemia. But in principle, the approach is, of course, suitable also for other blood cancers. There is no fundamental difference. So we may very well have the ability to expand usage beyond.
In addition to blood cancer, we are also investigating the use of ATIR to treat patients with inherited blood disorders like thalassemia, for which there is currently no cure and patients are dependent on blood transfusions. Thalassemia patients lack the ability to produce functioning red blood cells, so if you replace the diseased bone marrow, then the new, healthy bone marrow could produce functioning red blood cells and the patient, in principle, could be cured.
What are your plans to get to the market?
We have completed Phase II with ATIR101 for blood cancer and initiated the Phase III. We have approval from a number of countries to start the study and we are now lining up sites in the US, Europe and Canada. In Europe, we filed for EMA approval a couple of weeks ago, at the end of April, based on positive Phase II data. We would expect a final “go or no go” from the EMA in the second half of 2018, which should allow us to launch the product in the EU in 2019.

How does the competition look like?
For the patients for whom a matched donor is not available, there are two approaches. One is the Baltimore protocol, currently used in the clinic, in which patients get transplanted with the full half-matched graft from the donor, immediately causing GvHD. The GvHD is then treated with high dose chemo within a couple of days after the transplant.
An alternative approach, still under development, is the T-cell depleted approach of Kiadis, Bellicum Pharmaceuticals and MolMed. What Bellicum and MolMed do is first transplant safe stem cells and then a separate dose of T-cells after about a month, just like we do. The fundamental difference is that they give all the T-cells, including those which could cause GvHD, after genetically engineering them with a suicide switch. If GvHD occurs, they infuse the suicide agent, which then kills all the T-cells.
The big question with that approach is, do you not also kill those T-cells that can protect against relapse and infection? We believe we have a superior alternative to the Baltimore protocol and to Bellicum and MolMed.
With Kiadis and many other companies advancing rapidly, what’s your view on how biotech is evolving in the Netherlands?
When I entered the biotech world in 2001, there were very few biotech companies and they were all very early stage. Crucell, in which I was a part of the management committee, was for a long time the largest and most successful company. When it was acquired by Johnson & Johnson in 2011 for €2.1Bn, we were the biggest at the time.
We’re now six years later, and I’m really happy to see that other biotechs have progressed well beyond where we were at the time with Crucell. With companies like Genmab and Galapagos having a product in or very near to the market and with much higher valuations. With companies like Acerta and Dezima, having gone from the bottom up and being acquired for a huge amount by pharma companies.
Within the more early stage in Utrecht, Leiden and Amsterdam, there are companies like Merus and ProQR. I think the biotech scene is really maturing and growing and that really helps, because there are just many more people now with relevant experience to help the growth of this sector.

And why do you think is that?
Well, I think there’s probably two main drivers for the Dutch biotech scene. One is just very good science coming out of the universities in the Netherlands. It’s also the Dutch entrepreneurial spirit. There were many situations where many of the biotech companies had a difficult time and it was often a real business sense and entrepreneurial spirit and drive to get medicines to patients that made companies persevere and end up being successful.
Galapagos is now doing absolutely great, but for a long time, their share price barely moved. But they were able to continue because they had the wisdom to set up a revenue-generating service business. Genmab had a very rough time where they had to lay off many people and they just kept going. They both have now multi-billion valuations.
And if I look at Kiadis and ProQR, for example, ProQR was based on technology acquired out of the US and the product Kiadis is now developing is based on technology that came in through an acquisition in Canada. So it’s a combination of good science in the Netherlands but certainly also from entrepreneurial spirit and the ability to think large and look across our own borders.
Images via Kiadis Pharma; crystal light, Yasonia /Shutterstock





