Novel immunotherapies, like T cell therapy, may have shown recent successes but they also come with a number of setbacks, such as long timelines and serious adverse reactions. As CEO of the Dutch biotech Kiadis Pharma, Arthur Lahr has been leading the development of a technology that could overcome these limitations by using natural killer cells.
While cancer immunotherapies address a number of the shortcomings traditional treatments have, patient survival rates are still low for many cancers. For example, in lung cancer, the most common form of cancer worldwide, the five-year survival rate lies at only 17.8%.
“Although there has been lots of progress in cancer therapy over the last decades, unfortunately, many people still suffer from cancer relapse after treatment and die from cancer,” said Lahr. “Current treatments do not cure people yet. And quite frankly, I do not believe people that say the magic bullet is around the corner and we will soon eradicate cancer.”
With more than 20 years of experience in the biotech industry, Lahr believes that progress in cancer therapies is possible with time. However, the vast differences seen in patients with the same cancer diagnosis and the fact that many tumors can escape current immunotherapies means there is a long road ahead before the ‘magic bullet’ can be found.
Cancer’s complexity stands in the way

The key problem, explained Lahr, is the fact that there are many subtypes of cancer and factors that can drive cells to become cancerous. “[Cancer] is not just one disease. It has one external manifestation, uncontrolled growth, but the underlying biology is incredibly different from patient to patient and even within patients.”
“It is essentially a disease of the immune system. The immune system of most people is able to keep cancer in check for a long time… But in reality, the immune system is never perfect, and mutations occur and cells go wild.”
Many current cancer immunotherapies are specifically engineered to attack cancer in a way that would never occur in the human body naturally. For example, the two approved chimeric antigen receptor (CAR) T cell therapies on the market today, Kymriah and Yescarta. These are developed by engineering the patient’s own T cells to attack their B cells, which are the ones affected by their cancer.
While immunotherapies like CAR T are based on engineered T cells, the team at Kiadis uses natural killer (NK) cells to restore and boost the natural protection of the immune system. The company is developing therapies against blood cancers, such as acute myeloid leukemia, where survival rates are low and relapses occur frequently.
“We want to use the best that nature and the human immune system have to offer,” explained Lahr. “For this, NK cells are a very logical candidate. They are considered safer than T cells, and can be offered off-the-shelf and are available to patients as soon as they are needed.”
Leveraging the natural killing mechanisms of NK cells
As part of the innate immune system, NK cells are first in line to respond to pathogens or mutating cells. NK cells and T cells have very similar killing mechanisms, but they differ widely in how they detect danger.
For instance, T cells scan cells for signs of cancer or infection using a protein called a T cell receptor, whereas NK cells are equipped to pick up signs of stress in the form of specific membrane proteins, which often occur in infected or cancerous cells. So while T cells rely on the T cell receptor to identify cancerous cells or pathogens, NK cells can detect a much wider range of markers.
Moreover, NK cells do not cause cytokine release syndrome or graft versus host disease, which are common side effects of T cell therapy.
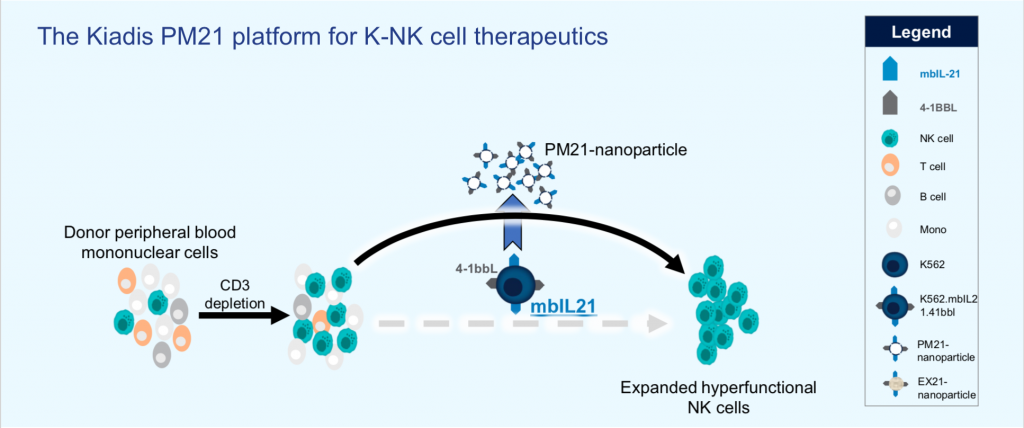
Leveraging the advantages of NK cells, the team at Kiadis has developed a nanoparticle-based technology that can supercharge the killing capacity of the NK cells in its cell therapy. Specifically, nanoparticles carrying selected membrane proteins can stimulate NK cells to rapidly expand, resulting in the generation of large numbers of highly active, cytotoxic NK cells.
To enhance their anti-tumor characteristics, many immunotherapies are being used or are under investigation as combination therapies. In July 2020, Kiadis signed a deal with Sanofi, in which its NK cell therapy will be used in combination with the company’s recently approved antibody therapy Sarclisa.
With a potential value of €875M, this collaboration aims to boost the immune system’s response in patients with multiple myeloma. While this deal focuses on blood cancer, Lahr is positive that his company’s NK cell therapy in combination with other immunotherapies can work in solid tumors as well. This would address one of the main challenges in current cancer immunotherapy, namely, getting immunotherapies to target and kill solid tumors.
“There are a number of studies with Herceptin and Erbitux that showed outcomes are just better in patients that have more potent NK cells… so if you combine our more potent NK cells with these products, you might see a substantial improvement in their benefit,” Lahr said.
NK cell therapy beyond cancer

Kiadis’ NK cell technology can not only be applied to cancer. As early as 2017, the company showed that NK cells are able to reduce viral loads in immunocompromised patients.
At the start of the Covid-19 pandemic, therefore, the team saw an opportunity to develop a pandemic preparedness platform on the basis of its NK cell technology. The platform focuses on immunocompromised patients, including the elderly, the recently transplanted, and patients with autoimmune diseases, who are taking immunosuppressants.
“These patients don’t respond well to the seasonal flu vaccine, so [they] are very much at risk,” Lahr explained. “And what we are creating is a universal product that can be given as an additional prophylaxis or treatment… to protect immunocompromised patients from flu, SARS, respiratory syncytial virus, and other viral infections. We want to develop a pandemic preparedness platform for future pandemics for any of these strains. That’s the ultimate goal of this program.”
Just recently, the FDA approved a phase I/II clinical trial that will test the effects of Kiadis’ off-the-shelf NK cell therapy platform in hospitalized patients with mild to moderate Covid-19. The research is being carried out in collaboration with the Ohio-based Abigail Wexner Research Institute and received €8M ($9.5M) in funding from the US government.
The key, Lahr told me, is to treat the patients early on in the progression of the disease, as NK cells are the immune system’s first line of defense. “It’s a very small therapeutic window and the aim is to reduce the initial viral load. The benefit of NK cells is that they not only reduce the initial viral load but they can also help accelerate the adaptive immune response. Ultimately, you want the adaptive immune response to deal with the virus by itself because that’s what NK cells do in nature.”
A booming industry
So while NK cell therapy is a relatively new approach, it has shown promising outcomes in both cancer and infectious diseases, and other companies are also working in the field.
French biotech Innate Pharma, for example, is developing an NK cell engager, which can bind to the surface of tumor cells and recruit and activate NK cells to cause tumor cell death.
And very recently, German biotech Affimed and California-based biotech NKMax America announced a collaboration to study Affimed’s innate cell engager in combination with NKMax America’s NK cell therapy in solid tumors.
The success of NK cells is also shown in recent investments in the field. For example, Irish biotech ONK Therapeutics, an off-the-shelf NK cell therapy company, recently raised €6.8M ($8M) from US-based investor Acorn Bioventures and its current shareholders. This brings the total raised by the company in the last six months to €12.4M ($14.6M).
As money pours in and interests are piqued, NK cells are changing the face of the immunotherapy field, and, without a doubt, we will see many more exciting developments in the future.
Cover image via Anastasiia Slynko, article images via E. Resko and Kiadis Pharma.





