Vaccines for COVID-19 have saved lives all over the world, but their protection is threatened by the rise of variants of concern. Companies are developing ways to combat the evolving virus, such as booster shots and universal coronavirus vaccines.
During 2021, a plethora of COVID-19 vaccines was rolled out around the world. Since then, mass vaccination campaigns with vaccines from AstraZeneca, Moderna, BioNTech/Pfizer and Johnson & Johnson have saved many lives.
However, we’ve also seen the rise of variants of the virus responsible for COVID-19, SARS-CoV-2. The highly infectious delta variant emerged in March 2021 to become the dominant form in many countries. It was then supplanted by omicron in late 2021, which branched into multiple subvariants in 2022, with BA.4 and BA.5 now in the driving seat. There could also be other variants of SARS-CoV-2 lurking in the population undetected by genomic screening.
First-generation vaccines provide strong protection from severe COVID-19 caused by many of the newer variants, but are limited in preventing their transmission. It’s also possible that another SARS-CoV-2 variant could emerge that can completely evade protection from our vaccine defenses.
The vaccine arms race
When the coronavirus SARS-CoV-2 reproduces itself in cells, errors often appear in the copying process. Many such errors are corrected or have no noticeable effects, but others can lead to changes in its structure and impact the way the virus infects humans.
“The more people infected with the virus, the more random mutations arise. Some of these mutations are beneficial to viral replication so they self-perpetuate,” said Thomas Rademacher, co-founder and CEO of the UK firm Emergex Vaccines. “It’s a game of chance that could be equated to rolling a die and getting a six. The more dice you roll, the higher the likelihood of getting a six.”
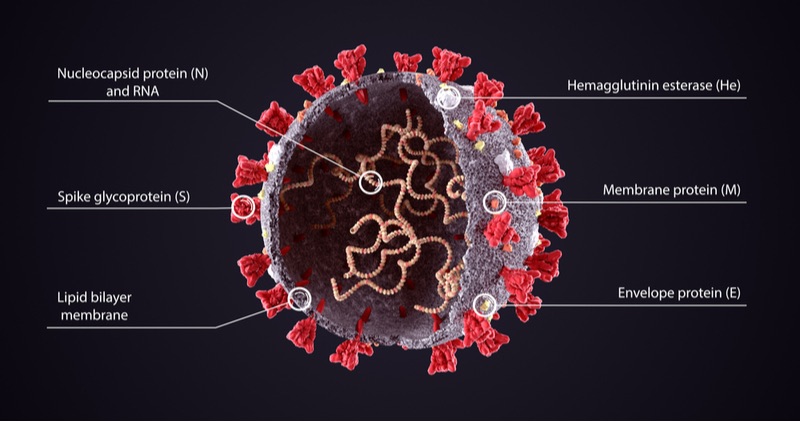
Most COVID-19 vaccines in the market and in development train the immune system to detect antigens on the surface of SARS-CoV-2. The most frequently used antigen is the spike protein, which is seen as the most effective vaccine target. However, mutations to the spike protein underlie the main variants causing concern.
“The more people becoming infected or vaccinated, the higher the selection pressure on the spike antigen becomes. This makes the key target of the antibody response more subject to mutations,” said Alexandre Le Vert, executive chairman and co-founder of the French vaccine firm Osivax.
While most wealthy nations have vaccinated the majority of their populations, the rollout of vaccinations remains very uneven in many low income countries. This gives the virus time to mutate its spike protein and escape the vaccine. In time, some vaccines could even start to exacerbate COVID-19 infections via a phenomenon known as antibody-dependent enhancement, where certain antibodies stick to the virus incorrectly and end up contributing to the infection.
“We should expect more variants to appear regularly and we will most likely reach a situation similar to flu, where multiple variants will be circulating every winter season,” warned Le Vert.
Booster shots of many first-generation vaccines were rolled out last year in many countries to combat renewed COVID-19 outbreaks. Additionally, big vaccine developers including Moderna and BioNTech/Pfizer are working on variant-specific booster versions of their vaccines. Moderna launched a phase 2 trial of an omicron-specific candidate in March 2022, while BioNTech and Pfizer began a phase 2 trial of their own omicron-targeted vaccine in July 2022. It’s unclear, however, what future variants could arise, and how much protection these vaccines could provide.
Enter the universal COVID-19 vaccine
One way to circumvent the vaccine arms race is developing a future-proof, universal coronavirus vaccine. Emergex sees a key clue in live attenuated vaccines, which use a weakened form of the disease-causing pathogen.
Many approved live attenuated vaccines have stayed effective for decades, such as for yellow fever. Emergex believes this is because they allow T cells, a key part of our immune ‘memory’ for future infections, to target proteins inside the virus rather than on the surface.
“Many of the vaccines currently being developed for COVID-19 are targeting surface proteins and particularly the spike [protein],” said Rademacher. “This may not produce an equivalently safe, effective and long-lived immune response compared to that seen with live attenuated vaccines.”
Despite their advantages, live attenuated vaccines are tricky to transport and may be unsafe for people with weakened immune systems. Most developers have opted for other approaches such as viral vectors, messenger RNA (mRNA) and inactivated vaccines, such as one recently marketed by Valneva.
In April 2022, Emergex’s variant-resistant COVID-19 vaccine was administered to 26 volunteers in an ongoing phase 1 trial. The synthetic vaccine mimics internal SARS-CoV-2 antigens and trains T cells to detect infected cells. The trial is expected to end later this year. This week, meanwhile, Emergex prepared the manufacturing process for a phase 1 trial of another universal vaccine: this time protecting against many forms of influenza.
Osivax, in turn, is developing a preclinical-stage vaccine candidate that consists of nanoparticles carrying copies of internal COVID-19 antigens. The company is bankrolling the vaccine’s development via a €32M funding round raised in July 2020. Osivax is also developing a universal vaccine candidate for influenza in ongoing phase 2a trials.
“We believe that targeting internal antigens such as the nucleocapsid presents an advantage over surface antigens as they have a much lower mutation rate,” said Le Vert. He added that an immune T cell response against these internal antigens could protect against both current and future strains of COVID-19.
The Belgian company eTheRNA is using an mRNA approach to make its own variant-resistant COVID-19 vaccine. Unlike BioNTech and Moderna’s mRNA vaccines, eTheRNA’s candidate targets a mix of SARS-CoV-2 antigens both on the surface and inside the virus. It also places more focus on activating T cells than current vaccines, which are primarily designed to stimulate the production of antibodies against the virus.
“The viral surface is more sensitive to mutation and strain drift – and an antibody-based approach is potentially vulnerable to such changes,” said Wim Tiest, eTheRNA’s director of infectious disease program strategy.
“That is why an approach like ours that targets a broader part of the virus and triggers a multi-angled immune response offers a valuable alternative despite the longer development timelines.”
The UK startup Baseimmune is deploying artificial intelligence (AI) to lead the way to a universal COVID-19 vaccine. With the help of a €4.2M investment in October 2021, Baseimmune aims to design synthetic antigens tailored by AI-guided algorithms to produce long-lasting immunity to viral variants. The antigens can be used in a variety of vaccine platforms including mRNA and viral vectors.
A versatile COVID-19 vaccine arsenal
Out of the hundreds of COVID-19 vaccines in development worldwide, only a minority target internal viral antigens. Other companies seem confident that vaccines against spike and other surface targets will protect over the long term, provided the proper booster shots are available.
“It is very difficult to actually predict when a ‘perfect’ escape variant will arise,” said Diane Van Hoorick, VP translational development at eTheRNA. “In response to mutational variants, next-generation vaccines with an alternative [spike] protein sequence could be implemented on an even shorter time track when building on the expertise gained already.”
Nevertheless, it seems prudent to build up a diverse range of vaccine technology that can provide long-term protection. Strengthening our defences against variants also has an added bonus: fewer vaccines are diverted to boosting an already vaccinated population and more can go to regions where they are needed.
“For novel variants, SARS-CoV-2 is a pathogen that mutates at a relatively low rate,” said Baseimmune’s co-founder and CSO, Ariane Gomes. “The more widespread use of vaccines and the growing numbers of individuals that have been exposed to the virus means that we have the tools to bring SARS-CoV-2 to a level of endemicity with localized and small outbreaks.”
Additionally, defenses will always be needed against pandemics in the future.
“The current situation highlights the need to move fast with diversity of new vaccines and new and adaptable vaccine technologies that have different courses of action, and that have the potential to deliver broader protection,” said Rademacher.
This article was originally published in 2020 and has been updated with the latest in COVID-19 vaccine research.