“Having access to all the chemistry and cell biology assays in context helps us to better plan for the next R&D stages.” — Gaston Habets, chief development officer, Opna Bio
Opna Bio is focused on the discovery and development of novel small molecule oncology therapeutics. Founded in 2019 in Lausanne, Switzerland, Opna is progressing a portfolio of early discovery to mid-clinical stage small molecule drug programs targeting the hallmarks of cancers. With multiple preclinical projects generating massive and diverse datasets, from compound structures and chemistry, to screening and biology assay workflows, Opna has established CDD Vault Registration System and ELN as the central repository for all its preclinical data.
Opna was initially set up to develop compounds against a novel oncology target, fragile X mental retardation protein (FMRP), which was discovered in the laboratory of Opna cofounder Douglas Hanahan at EPFL. In March 2022, Opna then acquired 5 small molecule programs from Plexxikon, following closure of the latter by parent company Daiichi Sankyo. Additional laboratory space in South San Francisco was acquired during the first half of 2022, and in November 2022 the company announced raising $38 million in Series A funding to support development of its preclinical and clinical pipelines.

Gaston Habets, Opna’s chief development officer, was recruited to Opna during August 2020 from Plexxikon, and is one of half a dozen scientists who joined Opna from Plexxikon over the last two years, including Opna CEO Gideon Bollag.
Habets heads the screening programs and explained that the requirement to set up a flexible environment for managing data was evident from an early stage in the company’s operation. “Even with just a single discovery project in our portfolio, there was an obvious need to establish a database platform for recording, managing, and analyzing data.”
Opna Bio needed an informatics solution
Opna was looking for a digital platform that could make it easier to find, view, analyze, and exchange biology and chemistry data within the company.
“One of the key requirements was to store and manage all of our basic biology data, from reporting on cell lines, to looking at gene expression profiles,” Habets noted.
“We wanted to have all of that documented in such a way that people wouldn’t have to ask where they could find data they needed, and that meant not relying on a traditional folder system. And then when we started our chemistry screening programs, we needed a platform that could store compound and chemistry data.”
Opna partners its screening programs with San Francisco-based R2M Pharma, a CRO that was already using CDD for managing and sharing data securely, with its partners.
“We’d witnessed CDD Vault in action through our collaboration with R2M, and realized it could be an ideal platform for us in house,” Habets said.
“In 2021 we took up CDD Vault as our data management solution for the FMRP discovery project. The Vault and its ELN are now also used as this central repository for all data relating to our acquired preclinical pipeline. We also have acquired a licence for certain R2M scientists to access data relevant to our collaborations with them.”
One platform for all data at Opna Bio
With the Plexxikon program acquisitions, Opna now has six pipeline projects, including two clinical-stage compounds, and four others at the preclinical stage. FMRP –the target on which Opna was founded – is overexpressed in multiple cancers, deletion of which creates an immune desert and triggers antitumor activity. The FMRP project is still at the hit finding phase.
Habets acknowledged, “It’s not often that companies are founded on a single target, for which there is so little chemistry information available and we’re not aware that anybody else has been working on FMRP in a cancer context.”
Opna has transferred key existing data from the preclinical FMRP and preclinical Plexxikon programs into the Vault, as well as newly derived data and results generated in-house from continued research.
“So all of the data generated by us at Opna, by our screening CROs, and other partners is held in the Vault,” Habets noted.
“Having access to all the chemistry and cell biology assays in context in that single environment helps us to analyze existing data, and better plan for the next R&D stages.”
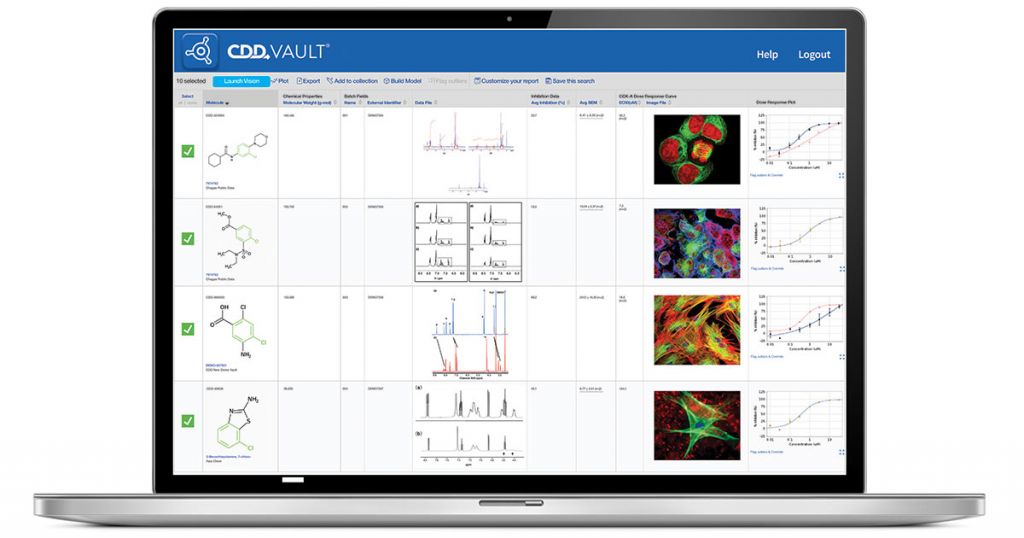
Opna Bio’s data management in action
In practice, Opna operates four Vaults.
“We have duplicates, so there’s a sandbox for each of our working Vaults,” Habets said.
“In brief, the bio/chemistry, or compound Vault is where we hold all the compound libraries that we have screened, and the compounds we synthesize. The ELN that goes with that holds all the screening data generated by testing these compounds. Our second Vault is a biology Vault, for which we have a discovery ELN that keeps data on all the cell lines and other biology. In fact the Vault itself carries an inventory of cell lines, proteins, antibodies, and reagents that we use or have purchased. And having two Vaults makes it possible to keep a detailed inventory for both chemistry and biology separately.”
Importantly, the Opna groups in Lausanne and San Francisco all have real-time access to the same system, so there’s no need to synchronize experimental results and compound structural data. The company’s scientists all view the same data generated at the two Opna sites and by the CROs, so geography is no barrier to data access or analysis. The one difference is that each site has its own inventory within the Vault.
“So if you have a cell line in Lausanne, then the San Francisco team can see it, but not make changes,” Habets pointed out.
Opna is a small, 15-strong team, with four scientists working in the biology lab in Lausanne, and 4 scientists based at the lab in San Francisco, which is leased from R2M.
“So there’s a lot of connectivity in our setup, both with respect to our programs, and our research sites,” Habets noted.
The cell biology and work with animal models is largely carried out at the Lausanne site, and where the team’s primary remit is the FMRP program.
The U.S. site carries out chemistry and biology for all FMRP and other programs, and employees there are also responsible for all the other pre- and clinical-stage projects.
“We have chemists in the US, and we also outsource our chemistry, as we are a small team.”
By offering a single platform for interpreting results and aiding decision making, CDD has played an integral role in supporting continued R&D progress for the preclinical programs. Support from the CDD team has also been a key enabler.
“Setting up the Vault was a process that involved working closely with the CDD team, and they have been supporting us as we moved forward,” Habets noted.
To learn more about CDD Vault and the many ways it can accelerate your drug discovery process, visit here.
Author: Sophia Ktori
Images: Shutterstock