One of the greatest challenges faced by researchers in preclinical oncology studies is the translatability of animal models. Mimicking human diseases accurately to investigate the effects of novel therapeutics is a demanding task. With the development of increasingly complex immunotherapies, biotech and pharma companies are looking for more relevant and accurate preclinical models to get the most out of their oncology research.

“The success rate in predicting clinical efficacy of anti-cancer modalities using current xenograft models has been reported to be only 30-40%,” says Dr. Rajendra Kumari, Global Head of Scientific Communication at Crown Bioscience. “These subcutaneous models have been used by oncology researchers to test the anti-tumor efficacy of their compounds for years. Although patient-derived xenograft models could be translated into human patients, for most therapeutics, especially immunotherapies, this is not necessarily the case.”
In order to recapitulate each cancer type more faithfully, researchers require models in which the tumor can interact with the complex microenvironment that is also found in humans. This relevant microenvironment can only be created in the organ where the tumor first developed.
Not only is a functional immune system required for the evaluation of immunotherapeutics, but researchers also need to have a thorough understanding of the disease biology and the limitations of the animal model for the proper interpretation of data.
Orthotopic vs. standard tumor models
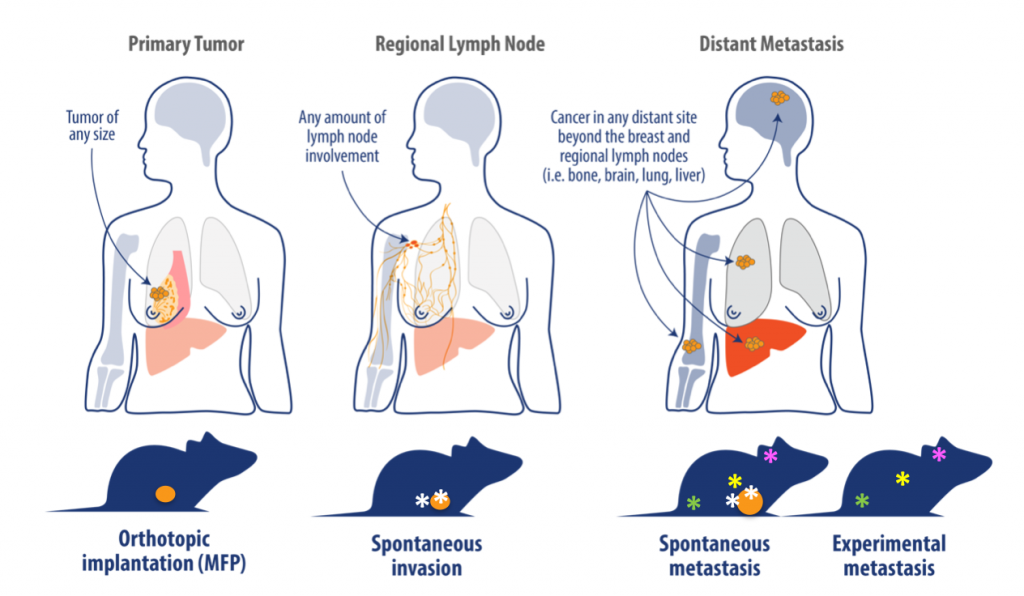
The growing demand for preclinical models that mirror human diseases more accurately, has led to the development of so-called orthotopic tumor models. “In orthotopic tumor models, tumor cells are engrafted into the relevant organ of tumor origin,” Kumari explains. “In an orthotopic breast cancer model, for example, breast cancer cells will be introduced into the mammary fat pad of female mice, prostate cancer cells into the prostate of male mice and so on.”
In standard subcutaneous models, by contrast, the tumor cells are engrafted under the skin, on the flank of the animal. Additionally, models such as GEMMs (genetically engineered mouse models), which spontaneously develop tumors within relevant organs due to disease-specific mutations, are not ideal for large-scale in vivo pharmacology studies.
Orthotopic tumor models are more clinically relevant than their subcutaneous counterparts, because of the establishment of an organ-specific tumor microenvironment which can differ in each cancer type. “The right microenvironment contains the relevant vasculature, a similar hypoxic condition as in the primary tumor, as well as stromal cell infiltration, including specific immune cells, and a greater inclination to metastasize,” Kumari explains.
“Orthotopic tumor implantation allows researchers to examine cancer progression more accurately and interrogate drugs more faithfully. Each organ or microenvironment is unique and cannot be replicated by subcutaneous implantation, so the growth of that particular tumor line will be influenced by the microenvironment and other cell types such as stromal cells and vasculature.”
The researchers at Crown Bioscience have further demonstrated that the infiltrating immune cells from subcutaneous mouse tumors differ from those in orthotopic tumors from the same model. “Our results suggest that the model type should be considered carefully when evaluating responses to immunotherapies in vivo,” Kumari explains. “Orthotopic tumor models have a greater potential to generate more patient-relevant pharmacodynamic profiles for immunotherapeutic agents, therefore providing a relevant efficacy and resistance profile.”
Challenges of orthotopic tumor models

While subcutaneous models are relatively easy to set up, the establishment of orthotopic tumor models demands specific surgical skills. This requires anesthesia, longer timelines, includes considerations of animal welfare, and the assessment of variability and reproducibility. Another major challenge is the tracking of tumor progression in orthotopic tumor models. Unlike subcutaneous models, tumors in orthotopic models are not always visible.
“The tumor progression in orthotopic models can be observed by frequent monitoring of clinical signs to establish humane endpoints or timed terminations to determine the duration of the model,” Kumari explains. “The quantification of tumor growth or the response to therapy, in the absence of a palpable tumor, is carried out at the final stage by histology, PCR, or other assays which can take weeks to read out.”
However, the monitoring of orthotopic tumors has become significantly more accessible thanks to the availability of cancer cell lines stably transduced to produce a bioluminescent signal. If the tumor is established using bioluminescent cells, it can be monitored by non-invasive imaging. Nevertheless, this requires anesthesia and specialized equipment. Moreover, the large amount of data derived from the imaging software needs experienced users for the downstream analysis.
The Crown Bioscience team has over 15 years of experience in orthotopic modeling and optical imaging. This has enabled them to develop a broad range of bioluminescent cell line derived models from solid tumors and liquid cancers, such as leukemia, and to effectively test anti-cancer agents in vivo using more clinically relevant models.
Using optical imaging to monitor disease progression
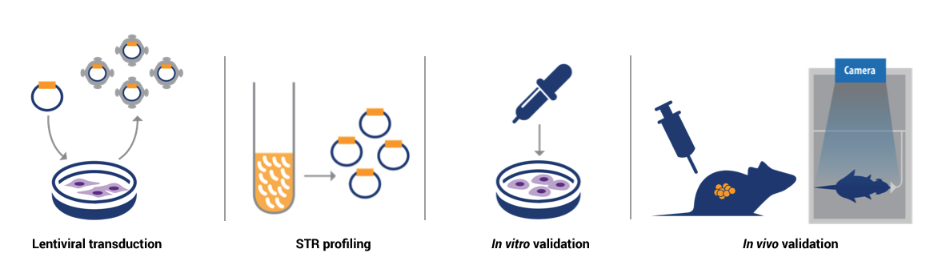
“Our model development efforts have opened up a wealth of possibilities for different cancer types, and we can now offer a range of models with an organ-specific tumor microenvironment, including spontaneous metastasis, experimental metastasis, as well as systemic models,” says Kumari. “Optical imaging allows the real-time monitoring of disease progression. Because of our expertise in this area we are able to execute preclinical studies effectively, with an in-depth understanding of what can be achieved and also build models to answer very specific questions.”
The team at Crown Bioscience has specialized in the application of optical imaging for longitudinal, real-time monitoring of orthotopic tumor burden. Tumor cells are engineered to stably express firefly luciferase, a protein responsible for generating a bioluminescent signal when exposed to its substrate. These bioluminescent cells are then engrafted into the organ-of-interest in the animal. In order to visualize the tumor, the substrate luciferin is administered to enable the biochemical reaction that emits light when the animals are imaged.
“The emitted signal is detected in real-time with a specialized imaging system and software, and quantified as a measure of tumor burden,” Kumari explains. “At CrownBio we specialize in the application of this technology for in vivo pharmacology studies. Essentially, we monitor tumor response to therapy in real-time by quantifying the bioluminescent signal emitted by the tumor within an animal.”
Optical imaging can be fast and efficient and has allowed the development of more refined study designs, which require fewer animals per treatment arm. Animals can be recruited at exactly the right time and monitored throughout the study, generating a vast amount of data. Compared to fluorescent imaging, bioluminescent imaging has a good signal-to-noise ratio, meaning the healthy tissue surrounding the tumor doesn’t interfere with the tumor itself when the signal is quantified.
“Bioluminescent optical imaging can be cost effective when you consider the high volume of data you can obtain from a smaller cohort of animals compared to standard tumor monitoring techniques,” Kumari says. “When combined with CT-imaging, tumor visualization can be represented in 3D in relation to the mouse anatomy, which provides further insight into tumor burden and invasion. Moreover, optical imaging can be used for other applications, such as monitoring drug biodistribution, delivery, and biological reporters.”
Want to learn more about Crown Bio’s work with orthotopic tumor models? Check out their new white paper or visit their website for more information!
Images via Shutterstock.com and Crown Bioscience
Author: Larissa Warneck, Science Journalist at Labiotech.eu