Adocia’s ready-to-use glucagon has proved to be safe in phase I trials. The company believes it can overthrow Novo Nordisk and Eli Lilly in rescuing severe hypoglycemia.
Based in Lyon, France, Adocia develops new formulations to improve the performance of diabetes drugs, including an ultra-rapid insulin to help type 1 diabetics control their blood glucose levels better. Today, the company has announced that its candidate BioChaperone Glucagon is as safe as Novo Nordisk’s commercial GlucaGen HypoKit.
Glucagon counteracts the effects of insulin, resulting in an increase in blood sugar levels. Thus, it’s used to treat cases of hypoglycemia, which can be life-threatening when blood sugar levels reach very low levels — something that can happen when a type 1 diabetic accidentally injects more insulin than they require.
However, current formulations of the hormone are not ready for immediate use. Commercial kits come in the form of a powder that has to be reconstituted into a solution through a process that takes between 7 and 9 steps. This can make it difficult for friends and family to administer it in case of a hypoglycemic emergency.
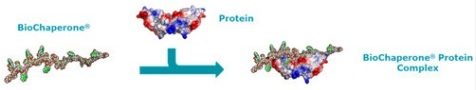
Adocia’s BioChaperone technology stabilizes glucagon in a water solution that can be injected directly to a patient in need. Therefore making the whole process much easier.
Still, when looking at the time it took to restore safe blood glucose’s levels, Adocia’s candidate took a median of 11 minutes, compared to 7 minutes for Novo Nordisk’s. A few minutes can be critical when treating severe hypoglycemia, but Adocia seems confident that its ready-to-use formulation carries bigger benefits.
The company cites a study in which only 13% of caregivers, and 0% of acquaintances without medical training, were able to inject a full dose when reconstituting Eli Lilly’s Glucagon kit. And those who managed to administer at least a partial dose still took around 2 minutes to do so.
Moving forward, Adocia is planning to give several applications to BioChaperone Glucagon beyond rescuing hypoglycemia. The drug is a great candidate to be used in dual artificial pancreas devices designed to automatically control the levels of blood sugar in diabetics, as well as a treatment for congenital hyperinsulinism and chronic hypoglycemia caused by bariatric weight loss surgery.
However, the French biotech is not the only one working on ready-to-use glucagon formulations. US-based Xeris Pharmaceuticals is testing its own glucagon rescue pen in Phase III, while Eli Lilly’s Locemia is developing a glucagon formulation delivered through the nose, which is getting ready for FDA approval.
Images via gala_gala /Shutterstock; Adocia