Erytech (France) is working on a drug-transportation system using red blood cells. Now US Rubius Therapeutics joins Erytech on this new approach to drug delivery, backed by the huge VC in the States; Flagship Ventures.
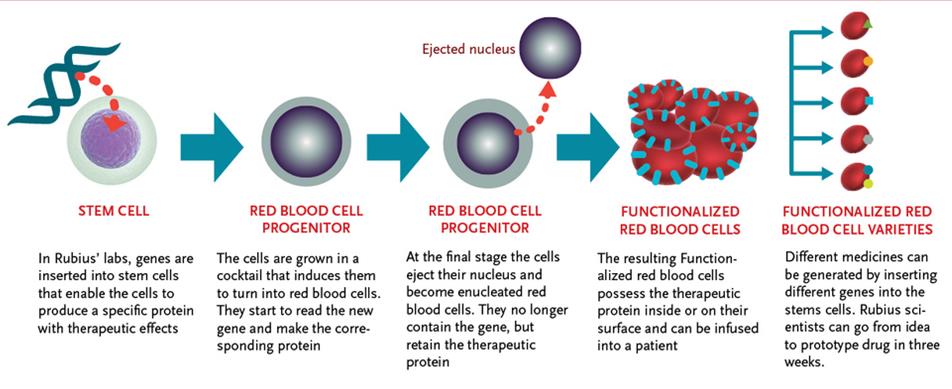
 Flagship Ventures has yet again shown its comprehensive portfolio in biotech through another EU inspired Biotech start-up. Rubius Therapeutics in Cambridge (Massachusetts) has been founded through Flagship’s VentureLabs to develop functionalized red blood cells for the treatment of autoimmune conditions, metabolic diseases, cancer, and other serious diseases.
Flagship Ventures has yet again shown its comprehensive portfolio in biotech through another EU inspired Biotech start-up. Rubius Therapeutics in Cambridge (Massachusetts) has been founded through Flagship’s VentureLabs to develop functionalized red blood cells for the treatment of autoimmune conditions, metabolic diseases, cancer, and other serious diseases.
Erytech in Lyon (France) have already developed a potential therapy for certain cancers using this method of drug delivery to a phase II trial stage. Rubius is therefore also pushing Red-Cell Therapeutics (RCTs), as a therapeutic approach to drug delivery.
Red blood cells are differentiated in bone marrow and the spleen and have a life-cycle of approximately 120 days, so understandably this is an appealing drug carrier in chronic disease given their longevity in the body. They also possess pharmacological advantages over other carriers due to their immunological familiarity within the patient.
One of Rubius’ scientific founders, Harvey Lodish, a professor of biology and bioengineering at MIT and a member of the Whitehead Institute, explained:
Red cells also have profound effects on the immune system and may ultimately transform the way we treat autoimmune diseases and allergies”
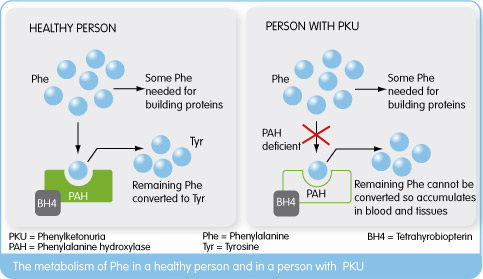
One of the primary conditions Rubius will be targeting is the super rare metabolic disorder Phenylketonuria (PKU). Phenylketonuria is a rare genetically conferred metabolic disease, where the amino acid phenylanaline (PHE) is not broken down properly.
This is due to a dysfunctional phenylalanine hydroxylase (PAH) enzyme resulting from a deficiency of the enzymes co-factor Tetrahydrobiopterin(BH4), leading to high PHE amino acid levels (due to lack of enzymatic function) which cause a wide range of psychological symptoms necessitating life-long dietary management.
This start-up focus is therefore particularly encouraging for patients of this rare disease considering that in September, Merck (MSD) decided to cease their R&D partnership with BioMarin on a PKU portfolio (Kuvan and the Phase II PKU drug Peg-Pal) to instead divert resources to immuno-oncology pipelines.
Rubius has developed and pre-clinically validated a Red-Cell Therapeutic capable of enzymatically reducing phenylalanine levels. By continually lowering phenylalanine levels in the bloodstream, RCT therapy promises to allow PKU patients to live normally without extremely restrictive diets.
Although it is early days, this could be the news patients are waiting to hear, particularly given the lack of clarity on Peg-pal development in the PKU community. Flagships’ VentureLabs clearly have an eye for niche demand in the biotech start-up scene.