Nano-sized vesicles that are generated naturally during bone formation have the potential to regenerate bone, teeth and cartilage.
Researchers from the University of Birmingham have mimicked the body’s natural healing process to repair damaged tissue, for example, bone. The study, which is published in Scientific Reports, has found that vesicles produced by stimulated cells outperform the current gold standard, BMP-2. Bone fractures place a heavy burden on healthcare systems across the world, with osteoporosis alone causing 8.9 million fractures each year. The group believe that this could be a new approach for the regeneration of bone, teeth and cartilage.
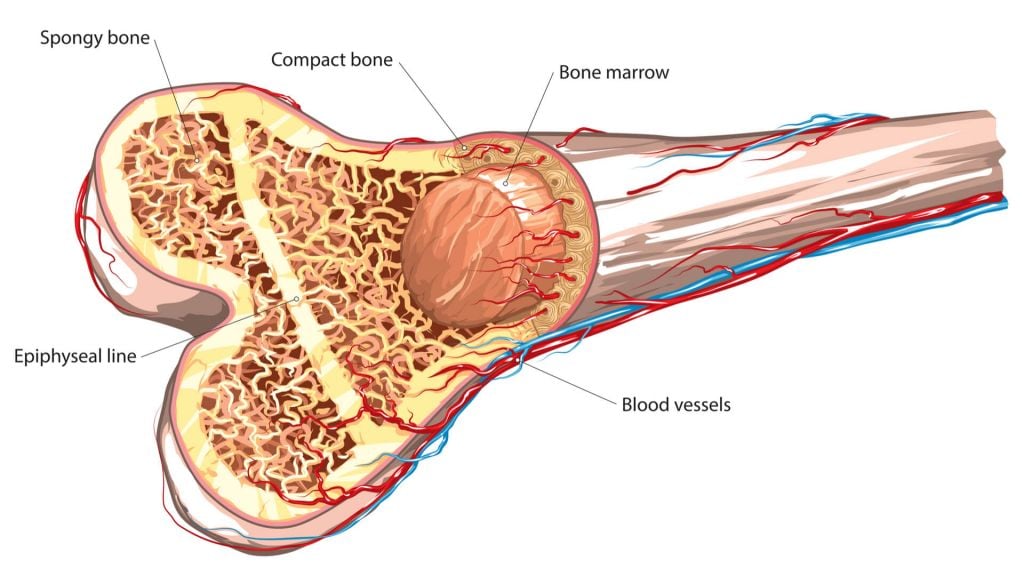
The technique uses extracellular vesicles derived from mineralizing osteoblasts – cells that synthesize bone – to enhance tissue regeneration. When these, and a source of phosphate, were added to mesenchymal stem cells, mineralization occurred indicating that the stem cells were heading towards forming bone. Impressively, the technique beat BMP-2 treatment, which uses a growth factor that is important to the development of bone and cartilage to stimulate repair.
Current approaches to repairing damaged bone, for example, autologous grafts, allogeneic bone and growth factor-based approaches have major drawbacks. A big advantage of the group’s approach is that it takes the benefits of cell-based therapies, without using the cells themselves, meaning it escapes the regulatory troubles that it might have otherwise faced.
It is no wonder that biotechs are scurrying to bring technology to the orthopedic devices market, with it expected to be worth $61B by 2023. Bonesupport is currently leading the charge to bring a product to the market to aid bone repair, with its Cerament technology awarded a CE mark in 2015. Bone Therapeutics’ ALLOB candidate is undergoing Phase I/IIa studies to tests its ability to treat severe osteoporosis.
Back in the lab, Austrian researchers have produced screws from recycled human bone to repair broken bones. Despite overcoming big problems associated with metal screws, surgery would remain a distressing prospect for patients. The research from the UK could offer a less invasive solution.
The approach developed in Birmingham, UK, ticks a lot of boxes for the successful regeneration of bone – it produces natural bone that will integrate into its environment and does this without the need for dangerous growth factors. The problem is ensuring that these very early but promising results can be transferred into animal models, and then into humans. If the group can manage that, it could cause quite a stir.
Images – dnaveh, NoPainNoGain / shutterstock.com