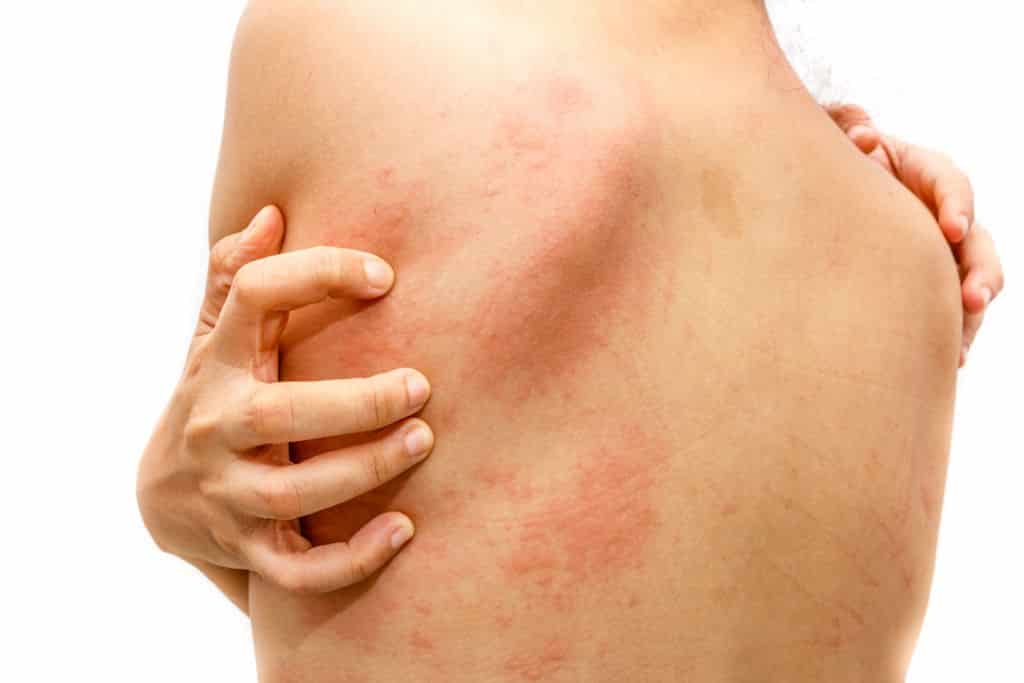
The U.S. Food and Drug Administration (FDA) has approved Dupixent (dupilumab) for the treatment of adult patients with prurigo nodularis.
Dupilumab is being jointly developed by Sanofi and Regeneron under a global collaboration agreement. To date, dupilumab has been studied across more than 60 clinical trials involving more than 10,000 patients with various chronic diseases driven in part by type 2 inflammation.
About 75,000 adults in the U.S. are living with the chronic skin condition and in need of new treatment options.
Dupixent under priority review: prurigo nodularis
The approval represents the second dermatology indication for Dupixent and fifth disease indication overall in the U.S. With this, Dupixent becomes the first and only medicine specifically indicated to treat the disease.
Prurigo nodularis is a debilitating skin disease with underlying type 2 inflammation and its impact on quality of life is one of the highest among inflammatory skin diseases. The FDA evaluated the Dupixent application for prurigo nodularis under Priority Review, which is granted to therapies that have the potential to provide significant improvements in the treatment, diagnosis or prevention of serious conditions.
Naimish Patel, head of global development, immunology and inflammation, at Sanofi, said: “Until today, there were limited treatment options to manage the relentless itch and associated sensations of burning and stinging skin that can negatively impact the lives of patients struggling with prurigo nodularis.
“Dupixent has the potential to transform the standard-of-care for prurigo nodularis patients by alleviating the key hallmarks of the disease, such as reducing itch and achieving clearer skin. With Dupixent now approved in two diseases in dermatology where type 2 inflammation is a central driver, we look forward to further evaluating the potential of inhibiting IL-4 and IL-13 in other chronic skin diseases.”
Phase 3 trials of Dupixent: skin condition
The FDA approval is based on data from two phase 3 trials, PRIME and PRIME2, evaluating the efficacy and safety of Dupixent in adults with the disease. Efficacy in these trials assessed the proportion of subjects with clinically meaningful reduction in itch, clearing of skin, or both.
The PRIME and PRIME2 phase 3 double-blind, placebo-controlled trials evaluated the efficacy and safety of Dupixent in 311 adults with uncontrolled prurigo nodularis.
About three times as many Dupixent patients (60% and 58%) experienced a clinically meaningful reduction in itch from baseline at 24 weeks, compared to 18% and 20% for placebo, the primary endpoint in PRIME.
Reduction in itch
44% and 37% of Dupixent patients experienced a clinically meaningful reduction in itch from baseline at 12 weeks, compared to 16% and 22% for placebo, the primary endpoint in PRIME2.
More than twice as many Dupixent patients (48% and 45%) achieved clear or almost clear skin at 24 weeks, compared to 18% and 16% for placebo.
More than three times as many Dupixent patients (39% and 32%) experienced both a clinically meaningful reduction in itch and clear or almost clear skin, compared to 9% and 9% of placebo patients at 24 weeks.
The safety results of the trial were generally consistent with the known safety profile of Dupixent in its approved dermatology indication.
Type 2 inflammation
George Yancopoulos, president and chief scientific officer at Regeneron, said: “Patients living with prurigo nodularis must often contend with dozens, if not hundreds, of itchy and painful nodules covering their body and have not had an approved treatment option for their disease.
“Dupixent has already transformed the treatment landscape of several diseases driven by type 2 inflammation – including atopic dermatitis, asthma, chronic rhinosinusitis with nasal polyposis and eosinophilic esophagitis – and been prescribed to more than half a million patients around the world for its approved indications. With this approval, those suffering with prurigo nodularis finally have a medicine to address the debilitating signs and symptoms of the disease.”
A regulatory filing for prurigo nodularis is under review by the European Medicines Agency, and submissions to regulatory authorities in additional countries are also planned in 2022.
Earlier this month, Dupixent had positive results from a phase 3 trial in children aged 6 months to five years old with uncontrolled moderate-to-severe dermatitis published.