Newsletter Signup - Under Article / In Page
"*" indicates required fields
Immatics and Amgen have inked a deal to develop bispecific drug candidates for cancer that could reach over €1B in milestones.
Immatics, an immuno-oncology biotech in Tübingen, Germany, has signed an agreement with biotech giant Amgen. Immatics will receive €28.5M ($30M) upfront and up to €475M ($500M) per program and Amgen will be responsible for clinical development, manufacturing and commercialization worldwide.
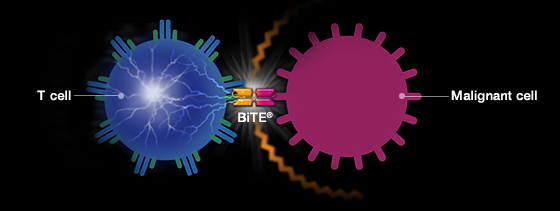
The collaboration will explore the development of novel cancer drugs by combining Immatics’ XPRESIDENT target discovery platform with Amgen’s Bispecific T-cell Engager (BiTE) technology. The team plans to build bispecific drug candidates that simultaneously bind an intracellular cancer antigen and a T-cell activator, signaling the immune cell to attack the tumor.
Immatics’ XPRESIDENT technology is, according to the company, the only high-throughput platform to identify, quantitate and prioritize tumor antigens. Unlike CAR-T therapies, it can target intracellular molecules, which account for 90% of tumor antigens, and solid tumors instead of only blood cancer.
Amgen already has a BiTE therapy in the market, Blincyto (blinatumomab), which hit the market in 2014 as a treatment for a rare form of acute lymphoblastic leukemia (ALL). New indications for the drug have been approved since then and its sales have been rapidly increasing.
The success of Blincyto seems to keep Amgen’s interest in launching more drugs using the same platform. The biotech giant currently has two BiTE candidates in Phase I clinical trials: AMG 211 for gastrointestinal adenocarcinoma and AMG 330 for acute myeloid leukemia (AML). Now, the incorporation of Immatics’ technology could boost Amgen’s platform and provide better treatments for cancer patients.
Images from Amgen
Oncology R&D trends and breakthrough innovations