Using the body’s own tools to eradicate cancers has been a dream of Medicine for quite some time. How did the field of immuno-oncology evolve into today’s Biotech hype?
 The near-miraculous results of immuno-oncology cell therapies have dominated news about cancer breakthroughs. Indeed, CAR-T is a game-changer and are at the forefront of the ways Biotech changed cancer treatments.
The near-miraculous results of immuno-oncology cell therapies have dominated news about cancer breakthroughs. Indeed, CAR-T is a game-changer and are at the forefront of the ways Biotech changed cancer treatments.
In a special feature, the New York Times traced back the origins of these wonder-therapies. It tells the story of a nearly 50-years quest to turbocharge patients’ immune systems against tumors, starting as an academic dream and ending up at the forefront of Biotechs’ pipelines.
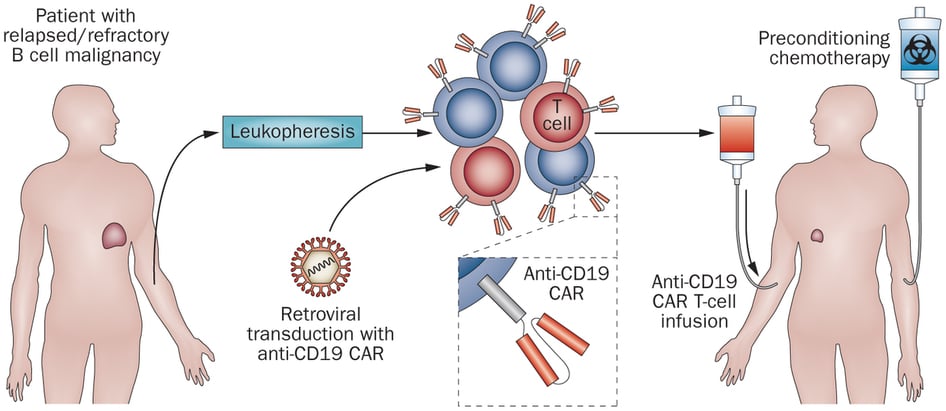
[In the 1990s and early 2000s] cancer cell therapy was still mostly an academic exercise; it was highly uncertain whether it would ever really work.
The article focuses mainly on T-cell therapies against cancer – an example of cutting-edge work in immuno-oncology.
More traditional approaches to immuno-oncology are monoclonal antibodies, a part of the immune system, to create a targeted therapy against cancer. With its 3 antibody-based blockbuster drugs, Roche has led in cancer treatments. Then there’s checkpoint blocking, a key strategy in immunotherapy.
However, some immuno-oncology researchers were fascinated with another component – the T-cell, the “soldier of the immune system“. What if they could boost its action against cancer?
Early lines of research did not engineer the cells themselves. Instead, it tried to maximize its action – a strategy still followed by Biotechs like TC Biopharma (Scotland).
At the National Cancer Institute (US), Steven Rosenberg tried giving patients T-cells harvested from pigs, administering interleukin-2 (which stimulates T-cell proliferation) and collecting tumor-infiltrating lymphocytes. All had very limited success, at best.
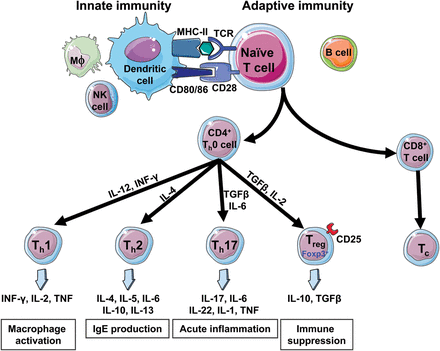
Meanwhile, the strategy of engineering cells was taking shape. Carl June was working in genetic engineering of T-cells to target HIV before his focus turned to cancer. He would later join the University of Pennsylvania.
Other scientists started engineering complex constructs that would allow T-cells to detect and kill cancer cells. These are the chimeric antigen receptor (CAR). However…
Pharmaceutical companies showed little interest, preferring mass-produced drugs, one size fits all, rather than a treatment that would be made separately for each patient.
Of course, this changed with the first clinical results, which showed incredible remissions. The researchers went to form partnerships with big Pharma and Biotechs that are now leading the CAR-T field, such as Novartis (with University of Pennsylvania), Kite Pharma (with the National Cancer Institute) and Juno Therapeutics (with Sloan Kettering).
And what’s next? Well, a slightly different approach, TCR, could broaden target possibilities – Adaptimmune is one of the Biotechs working in this type of therapies. Then, there’s the off-the-shelf strategy for CAR-T of Cellectis and engineered switches to control the therapy. Hopefully, this can bring CAR-T to more types of cancer. After all, despite all progress, T-cell therapies are still far to be the absolute cure for cancer.
If you are interested in the development of T-cell therapies, be sure to check the original article in the New York Times. (Philip called it a masterpiece ;))
Feature Image Credit: ‘Cancer Cells’ © Vitanovski (BigStock ID81969485)
Figure 1 Credit: Klebanoff et al. (2014) . Nature Reviews Clinical Oncology (doi: 10.1038/nrclinonc.2014.190)
Figure 2 Credit: Idris-Khodja et al. (2014) Dual opposing roles of adaptive immunity in hypertension. European Heart Journal (doi: 10.1093/eurheartj/ehu119)