In our first part of the interview with Dr David Hallett, Executive Vice President at Evotec, we discussed the uniqueness of Induced Pluripotent Stem Cells (iPS Cells), novel developments occurring in iPS Cell research, and the challenges researchers face.
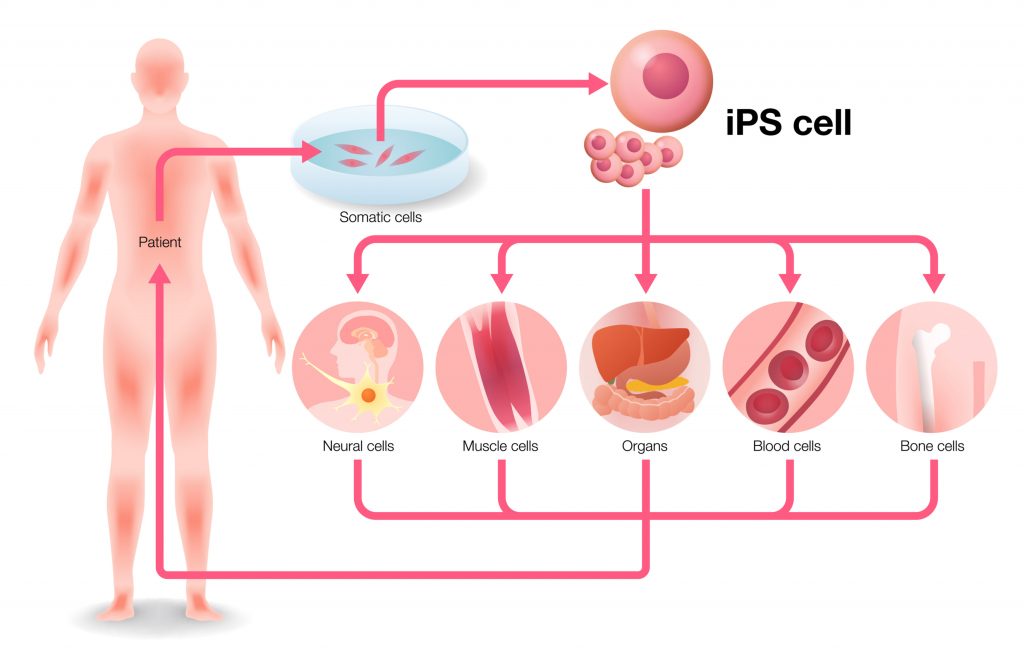
Let us have a short recap of what we have learned so far: iPS Cells were first discovered by Shinya Yamanaka and his colleagues in 2006. The researchers introduced specific transcription factors into mature adult cells, converting them back into a pluripotent state, and resulting in their ability to differentiate into any type of cell found in the human body.
Apart from this unique characteristic of being pluripotent, iPS Cells can also be maintained in culture and multiplied in a very short period of time. A further benefit is the fact that the reprogramming step can be executed using easily accessible tissues such as skin and blood cells, so there is no need for invasive procedures.

We also learnt that there are two primary applications for iPS Cells. One is in cell replacement therapy, where, for example, iPS Cells can be differentiated into pancreatic beta cells in the case of diabetes, or dopaminergic neurons in the case of Parkinson’s disease, and then be transplanted back into patients to replace their own defective cells. The second is in the broader field of drug discovery. Here, iPS cells are being used to explore the underlying causes of human diseases at a molecular level.
In the first part of the interview with Dave, he described the vast scientific background of iPS Cells. This time, he has given us an insight into why collaborating with other companies is essential in the field of iPS Cell research. We delve into Evotec’s partnerships and their research, and discover the future of iPS Cells and their implications for humankind.
Dave, what players are involved in the iPS Cell landscape and what is the importance of partnerships in this field?

In the first part of the interview, I gave you a sense of how broad this field is and the many components that are needed to help discover and develop novel therapeutics: sourcing patient-derived material, reprogramming, controlled differentiation, gene editing, disease modelling, phenotypic screening, cell transplantation and cell delivery technologies.
One company alone cannot handle this – it is just too big an area. Partnerships provide access to established technologies so that a single company does not have to build everything internally. It can also enable a pooling of resources, accelerating projects with both partners sharing the commercial risks and rewards.

Evotec’s collaboration with Sanofi, for example, has allowed both parties to explore a cell therapy option for Type 1 diabetes. Sanofi benefits from Evotec’s multi-year investment into the robust and reproducible production of human beta cells, whilst Evotec profits from Sanofi’s manufacturing expertise and technology network required in the evaluation and production of encapsulation devices and cells. Having a partner with the scale and resources of Sanofi accelerates the search for a potential cure.
There are numerous commercial, academic, not-for-profit and consortia that help enable access to technologies or tools, as well as performing discovery and clinical research. The sourcing of patient derived iPS Cells for instance. Evotec collaborates with Censo Biotechnologies to gain access to their deep knowledge of and infrastructure in reprogramming techniques.
In a similar manner, the European Bank for induced pluripotent Stem Cells (EBiSC) is a not-for-profit iPS Cell bank providing researchers across academia and industry with access to quality-controlled, disease-relevant, research-grade iPS Cell lines, data and cell services.
Similar groups exist in the United States – the California Institute for Regenerative Medicine has made huge investments over the last decade with the goal of accelerating the discovery and development of new therapies.
Dave, how does Evotec apply iPS Cell technology to drug discovery?
Evotec is a company with the goal of identifying novel drug targets and developing breakthrough therapeutics with our partners. We apply our technology platforms to evaluate patient-derived iPS Cells and other cell types, building disease-relevant assays that can then be used to screen small molecules and other therapeutic modalities.
We also have a substantial infrastructure and capabilities to optimize screening hits through to early clinical development. By using patient derived iPS Cells and through careful selection of the in vitro assays we hope see much better translation when molecules enter the clinic.
Evotec has paired up with Sanofi and Celgene to work on diabetes and neurodegenerative diseases, respectively. Can you tell us more about these projects and comment on the advantages in using iPS Cell technology?
Several years ago, we entered into a collaboration with Sanofi that focuses on diabetes. Within this partnership, we have taken human pluripotent cells and developed a differentiation protocol to deliver functional human beta cells in a robust and reproducible manner. We continue to work on upscaling and further refinement towards a large-scale process.

With Celgene, we are working in the field of neurodegeneration. We are building a broad collection of patient-derived iPS Cells and isogenic controls covering diseases such as ALS, Parkinson’s, Alzheimer’s and Huntington’s.
We have developed differentiation protocols to generate relevant neuronal cell types and we are investigating the cellular effects of the various (genetic) mutations to understand dysregulated pathways in these diseases. Then we develop screening assays using these neurons to which we apply compound collections from both Celgene and Evotec. Ultimately, we aim to deliver compounds to the clinic that can slow the progression or even cure a disease.
What are some of the challenges that you and others have identified and what novel research is taking place to address these?
The research community continues to invest heavily into the uses of iPS Cells and their derivatives. Several groups are looking at how to artificially age cells. Why? When you differentiate and mature iPS Cells in culture, for example to produce neurons, the cells resemble those found in embryos, as they are perhaps 4-8 weeks old when we use them.
Now, consider the study of diseases that are typically adult-onset, such as Alzheimer’s disease or ALS. The affected neurons in these cases might be 40 to 70 years old. Clearly, researchers would prefer to study this disease in a system that more closely resembles the ageing adult brain.
Another area of intense research is that of multi-cellular systems, called organoids. Much of the work we are currently doing examines a single cell type in vitro, like human beta cells, human podocytes or human forebrain neurons.
This is clearly very reductionist. In disease, whole systems are typically affected. Organs, such as the kidney contain many different cell types, as does the brain. We need to generate “organs in a dish” if we want to study complex systems. Several groups are making great progress in this arena, for instance “mini-brains” and artificial retinas, but they currently take months to generate and in most cases are still not reproducible enough for routine use.
Tell us, what is the Future of iPS Cells and what could that mean for humankind?
I am optimistic that in the very near future scientists will be able to routinely access the majority of human cell types derived from iPS Cells from commercial sources. This will be done at costs that may eliminate the use of commonly used cell types such as HEK, cells derived from human embryonic kidney cells, and CHO, Chinese hamster ovary cells.
Couple this with the large biorepositories that are assembling collections of patient-derived iPS Cells and we create a virtuous circle where everyone can easily access the tools required to study human disease in human cells.
Cell replacement therapy is also entering an important era. Progress in this area has been slow and continues to present challenges, but a small number of clinical studies are currently taking place with cell types derived from human pluripotent cells, such as retinal pigment epithelium. It remains to be seen how broadly applicable this approach will be.
Are there any other significant projects or partnerships in the pipeline?
At Evotec there are several ongoing research efforts that utilise iPS Cells and their derivatives. For example, we are exploring Lysosomal Storage Disorders – inherited diseases that are characterised by an abnormal build-up of various toxic materials in the body’s cells and whose symptoms typically manifest very early in life. We are also using different cell-types found in the eye to discover novel treatments for conditions such as Age-Related Macular Degeneration and glaucoma.
Are you interested in learning more about iPS Cells? Then check out our first interview with Dr David Hallett! Or check out Evotec’s website for more information. For the very curious, Dave has also suggested some of his favourite iPS Cell-reads, so give them a look:
- A Brand New Outlook on Induced Pluripotent Stem Cells (Labiotech, Part 1)
- Pluripotent stem cells in disease modelling and drug discovery (Nature)
- Induced pluripotent stem cell technology: a decade of progress (Nature)
Images via Shebeko, lmstockwork, unoL, Life science, memorisz, Atthapon Raksthaput, MriMan, Syda Productions/Shutterstock
Partnering 2030: The Biotech Perspective 2023






