Since both Alzheimer’s Disease and Parkinson’s combined affect up to 50 million people worldwide, it is no wonder this new wave of break-through milestones is getting the industry excited…
 The Central Nervous System (CNS) comprises many complex tissues and highly specialized cell types, mutation in which can cause an array of deadly and chronic diseases. Some of these have severe Motor-ability consequences (e.g. Huntington’s, Parkinson’s, Motor Neurone Disease and Multiple Sclerosis), whilst others relay more damage to the cognitive abilities of a sufferer (i.e. Alzheimer’s and Dementia).
The Central Nervous System (CNS) comprises many complex tissues and highly specialized cell types, mutation in which can cause an array of deadly and chronic diseases. Some of these have severe Motor-ability consequences (e.g. Huntington’s, Parkinson’s, Motor Neurone Disease and Multiple Sclerosis), whilst others relay more damage to the cognitive abilities of a sufferer (i.e. Alzheimer’s and Dementia).
After many years of disappointment in the field, this year has proven to yield some major successes in Neurodegenerative disease, which is now experiencing a huge revival in the Biotech industry. Novel ideas and research areas in the CNS have been piling up Alzheimer’s, Parkinson’s and Huntington’s success-stories throughout 2015…

Pursuit of the Holy Grail in Alzheimer’s
Alzheimer’s destroys cognitive function over-time, with confusion and memory-loss leading to intense emotional distress and loss of independence. We are now in the midst of a Dementia epidemic, which worldwide governing bodies predict will eventually cripple the Western economy if a solution isn’t found soon. Hence the launch of the Dementia Discovery Fund by the World Health Organization, UK Government and an ‘A-Team’ of Biotech Giants (such as Eli Lilly, GSK, Biogen and more).

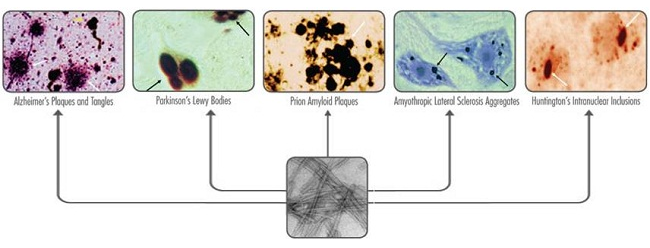
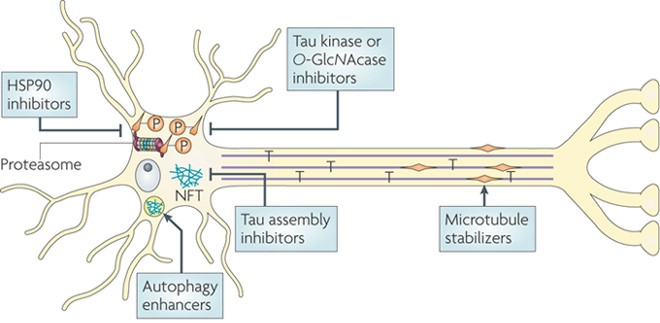
The pathophysiology behind Alzheimer’s and its associated dementia includes the build-up of the Beta-amyloid protein plaque (Aβ). The Aβ-plaque then create malformations of the Tau-protein, which aggregates into ‘tangles’. These tau-tangles have further downstream cytotoxic effects, creating irreparable plaques and damage.
Such diseases involving the tau-protein are therefore called ‘taupathies‘ and include rarer diseases like progressive supranuclear palsy (PSP). However, generally, all of the Neurodegenerative diseases discussed have protein malformations implicated in neuronal damage, differing by the particular protein and location of these ‘tangles’.
So, what exactly has happened this year in Alzheimer’s Research?
We’ve already covered the basic pharmaceutical arsenal for Alzheimer’s in our March review, but recent news has obviously since improved upon these drug targets. Just last month Nestle announced a new research partnership with AC Immune, for their Tau-targeting antibody candidate. AC Immune have also had significant interest from the Roche subsidiary Genentech, with a €384M investment milestone contract which was initiated back in 2012.
Other interesting developments include a phase I vaccine against Tau-tangles from Slovakian AXON Neuroscience, and now Switzerland’s AsceNeuron (originally a spin-off of Merck-Serono) having attracted €27.5M from French Sofinnova. A really helpful overview of TAI candidates for Alzheimer’s can be found here published in Biochemical Pharmacology 2014.
And More..?!
In August, phase III efficacy trials for Biogen‘s Aβ-plaque targeting monoclonal antibody candidate (Aducanumab) began, with phase I trials being launched in Japan too. Originally developed by Neurimmune in Schlieren (Switzerland), this phase III trial is set to run until 2022 in over 150 centres across the world.
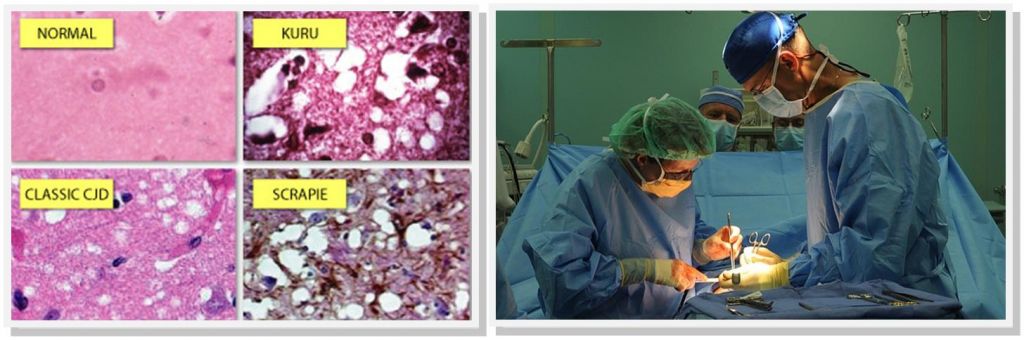
Another new (and still controversial) field for therapeutic research is the link between prion proteins and Aβ-plaques in Neurodegenerative disease, for which there has been increasing evidence of biochemical cross-over in Alzheimer’s and Parkinson’s. Prions are protein ‘malformations’, but are self-replicating and therefore responsible for the dementia in Creutzfeldt-Jakob’s disease and the infamous Cannibalism-associated Kuru Syndrome (which has a fascinating – if tragic – history).
What about Contagious Alzheimer’s?
Now, new research at the MRC Prion Unit at UCL (UK) has accused the transmission of prions through surgical instruments as having links to diseases such as Alzheimer’s – all of which is still hotly debated (and a potentially dangerous headline to put out there just yet)…as discussed in AAAS.
Takeda Cambridge (the UK subsidiary of global biotech Takeda Pharmaceuticals) will also collaborate with UCL’s Institute for Neurology to search for novel gene targets with significance to CNS disorders like Alzheimer’s and Parkinson’s…
Parkinson’s: A Disease Made Famous by Celebrity
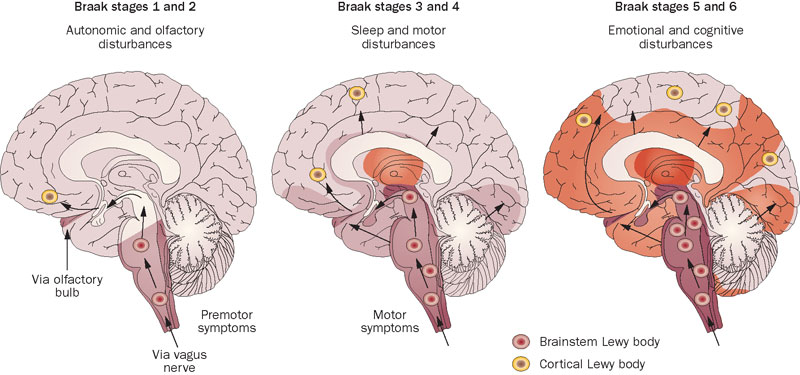
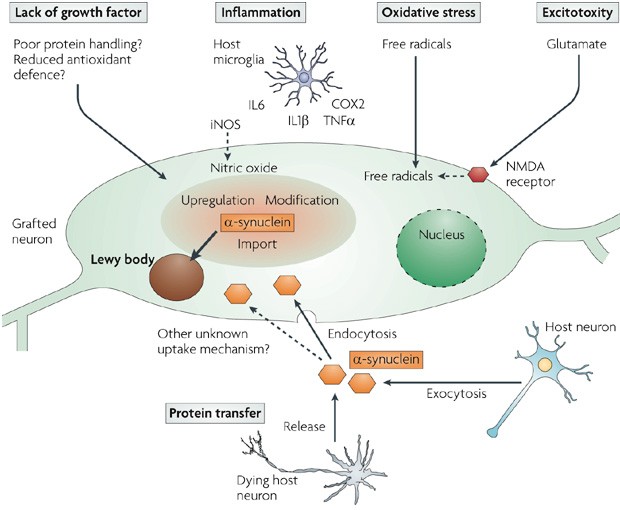
Characterized by movement disability and dementia, the gradual onset of PD is most commonly seen as tremors, which worsen over time. Famous people who have suffered Parkinson’s include the boxer Muhammed Ali, Johnny Cash and Pope John Paul II. Although still poorly understood, the accumulation of misfolded proteins aggregate into Lewy bodies which cause abnormally low levels of the neurotransmitter dopamine.
PD also has significant therapeutic overlap with Alzheimer’s (as discussed) due to the similarity in neurofibrillary ‘tangles’ of the tau protein compared to the alpha-synuclein protein in PD Lewy bodies.
Current treatments are aimed at replacing dopamine or mimicking its effects by chronically administering patients with the dopamine precursor L-DOPA, inhibitors of dopamine catabolic enzymes or direct dopamine receptor agonists. Instead, novel drug targets are those which provide more neuro-protection, as opposed to just temporary relief of the symptoms.
In February, the Swiss Biopharma Prexton secured €8.7M in a finance round (co-led by Sunstone Capital and Ysios Capital) for their phase I trial of a small molecule that targets the metabotropic Glutamate Receptor 4 (mGluR4). This is designed to prevent excess Glutamate (one proposed cause for Lewy body formation) and trials are due to start in 2016.
Other Swiss-related breakthroughs for the market came from the Italian biotechs Zambon and Newron, whose partnership over an L-DOPA add-on therapy, Xadago, received Swissmedic market approval just this month. As mentioned before, Swiss Neurimmune also has an anti-alpha-synuclein candidate (BIIB054) in its phase I/II.
In research, the FDA-approved anti-inflammatory drug Montelukast for asthma was unexpectedly found to be effective at improving neurodegenerative disease symptoms in rats, and helping memory loss caused by Lewy-Body Dementia. A team at the Paracelsus University in Salzburg (Austria) published these findings in Nature Communications last month, projecting potential future trials.
Finally, in spite of the negative outlook on new Parkinson’s drugs in the near future (although many pre-clinical candidates and ideas exist), the first-ever ‘Vaccine for Parkinson’s‘ by the Austrian biotech AFFiRiS in Vienna received an additional €10M financial backing in July. After positive phase I trial results for their PD01 vaccine last year, this anti-alpha-synuclein drug has received a lot of attention, including funding from the Michael J. Fox Foundation.
Here is a video by A. Demoulle Explaining the Affitope PD01 Vaccine Trial…
AFFiRiS also has a similar pre-clinical candidate for Huntington’s…
Huntington Holidays: A Premise of a Cure
Huntington’s is a heritable neurodegenerative disease which affects around 5-7 people per 100,000 (figures from the WHO). Caused by the autosomal dominant genetic mutation of the HTT gene, the result is neuronal damage by the toxic buildup of the mutant Huntington protein.
Being incurable, most treatments are palliative and look to delay the onset of neuromuscular spasms and devastating limitations on mobility.
Silencing the HTT Gene…
One way of targeting Huntington’s is inhibition of the HTT gene by using modified anti-sense oligonucleotides (ASO). These disrupt the mRNA translation of the toxic HTT protein, therefore creating a ‘Huntington Holiday Effect‘ – a therapeutic relief from the disease symptoms which has had great results in pre-clinical trials. A simplified but comprehensive review of ASOs in Neurodegenerative disease research can be read here.
One hugely exciting example includes the phase I/IIa trials announced by the San Diego (US) based Ionis Pharmaceuticals (previously known as Isis Pharmaceuticals) in partnership with Roche. Their small-molecule ISIS-HTT drug is one such ASO which targets the huntingtin mRNA.
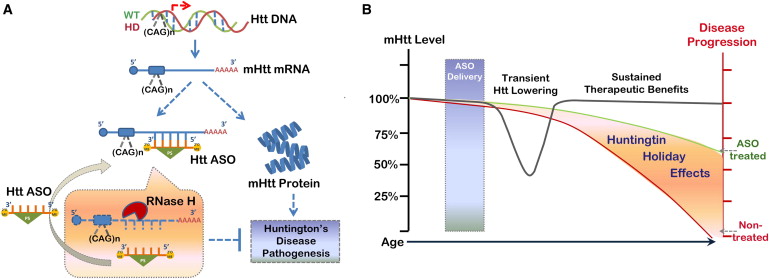
The trials are being led by Sarah Tabrizi, the director of the Huntington’s Disease Centre at UCL, and the ISIS-HTT candidate has also been granted Orphan drug status. On discussion of the ISIS trials, the chief executive of the Huntington’s Disease Association, Cath Stanley, told the BBC:
This is the first, potential, major breakthrough in terms of delaying symptoms of Huntington’s disease, it’s such an exciting step forward“
Whilst the aforementioned PBT2 failed to achieve its primary endpoint (to reduce beta-amyloid plaque levels) in the phase II trials for Alzheimer’s last year, the Australian spin-off from the University of Melbourne, Prana Bio, was also granted EMA Orphan Drug Designation for its indication in Huntington’s in July.
Their phase II results from the ‘Raise4HD’ trial last year were published in The Lancet Neurology in January.

So, of the tens of millions of people affected by these debilitating diseases, 2015 has proven a really exciting year for neurology medicine. In attempting to cover such a broad field, the general trend is that Neurodegenerative disease IS making a comeback in Biotech.
Whilst progress may be slower in certain indications compared to others, the overlap in the pathologies of these diseases means that progress in Alzheimer’s can also push through further progress in Parkinson’s, ALS or Huntington’s too. And this is what is exciting. All for one, and one for all!
P.S. It’s such a complex field, and there are always new projects going under the radar…do you know of any other trials or research worth mentioning? Please let me know in a comment!