Natural killer cells are becoming a hot topic in the booming cancer cell therapy scene, with the field’s latest big deal being a €640M licensing agreement between the French biotech Cellectis and US company Cytovia.
T-cell immunotherapies like CAR T-cell therapy have revolutionized cancer treatment in recent times. However, they have a significant disadvantage. The patient needs to get their immune T cells extracted, engineered to target the tumor, and infused again. This approach is costly and takes weeks — a luxury that many cancer patients can’t afford. Also, the cells extracted are often of variable quality, making it harder to produce an effective cell therapy.
The recent deal between Cytovia and Cellectis is one of many exploring a potential solution to this challenge: natural killer (NK) cells — a key part of the innate immune system.
Like T cells, NK cells can be engineered to better recognize a specific tumor. But they have a couple of advantages. One is that they can detect more chemical signals of tumors than T cells. Another is that they are less prone to attacking healthy tissue than T cells are. This means it’s possible to give patients NK cell therapies derived from healthy donors with less risk of the foreign cells attacking the patient’s body.
Paris-based Cellectis is a well-known developer of CAR T-cell immunotherapies, using gene editing tools such as TALEN to modify immune T cells into cancer-hunting machines. Its agreement with Cytovia allows the US biotech the rights to use Cellectis’ TALEN approach to turn donor stem cells into NK cell therapies, with the aim of entering clinical testing around 2022. In return, Cellectis can expect up to around €628M in developmental milestone payments and €12M in cash or as an equity stake in Cytovia if unspecified conditions aren’t met by the end of the year.
According to Daniel Teper, Chairman and CEO of Cytovia Therapeutics, this technology “will yield NK and CAR-NK treatments with improved potency, persistence, and safety for a variety of cancers, including solid tumors.”
The most advanced NK cell therapies are currently being tested in phase II trials to treat solid tumors and blood cancer. Some of the most prominent biotechs in the field include Bellicum Pharmaceuticals and NantKwest in the US, and XNK Therapeutics in Sweden.
Last year saw an increase in European and North American deals surrounding NK cell therapies in cancer. A major beneficiary of this boom was Kiadis Pharma in the Netherlands, which was acquired by Sanofi in November last year, just months after an €875M licensing deal with the same company. This year has already kicked off with a huge deal in the US: the NK cell therapy firm Artiva Therapeutics recently launched a collaboration worth up to €1.5B with the big pharma MSD.
Smaller European companies have also seen more deals in the last several months. This month, for example, the Swiss company Onward Therapeutics invested in the French NK cell therapy developer EMERCell, becoming the majority shareholder.
“This deal enables Onward to leap forward in developing a foothold in the field of immuno-oncology, and it may synergize with the bispecific antibody project that we recently licensed from Biomunex,” stated Grace Yeh, Chairman and CEO of Onward. “We envision a great prospect in exploiting this off-the-shelf NK cell therapy and proprietary antibodies as two pillars to build our next generation of product portfolio.”
Another deal announced this month was a collaboration between the Irish startup ONK Therapeutics and the UK blood cancer charity Anthony Nolan. ONK develops off-the-shelf NK cell therapies that are engineered to carry two types of cancer-busting proteins. The charity will secure ONK a supply of umbilical cord blood and cord-derived NK cells to boost the development of this technology.
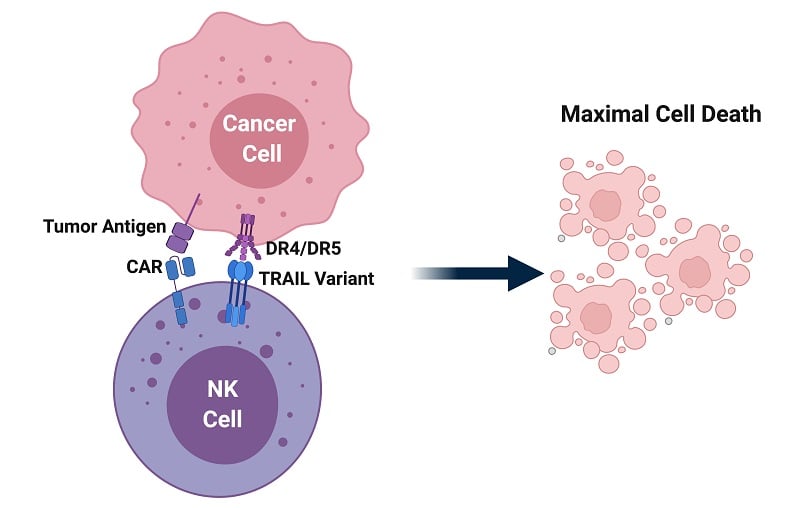
NK cells have shown a great potential for treating blood cancer, but their applications might not stop there. “One of the biggest challenges moving forward is how to build on the developments in the field and harness NK cell therapies for the treatment of solid cancers,” Anders Holm, COO and Head of Business Development of Oslo-based Zelluna Immunotherapy, told me.
In June last year, the Norwegian biotech raised €7.5M to tackle solid tumors using NK cells. To do this, the company is blending NK cells with another emerging technology based on T cell receptors (TCRs), which can detect cancer proteins that are normally hidden within the tumor cell.
“TCRs are a well-validated solid tumor targeting mechanism. When coupled with the killing capacity of NK cells, we believe this approach could be a highly effective and safe treatment option,” Holm explained.
One of the main bottlenecks in NK cell therapies is understanding the interactions between the NK cell and the target cancer cell. These interactions can determine the effectiveness of the treatment, as well as any potential undesired effects such as life-threatening levels of inflammation. Studying these interactions often takes a few days. However, Amsterdam-based LUMICKS offers a solution to shorten this time to 15 minutes.
“Our technology is based on applying acoustic forces at the single-cell level to probe the interaction strength between NK cells and their target cells, which are commonly tumor cells,” Rogier Reijmers, Principal Scientist at LUMICKS, told me. He added that performing these tests in a high-throughput manner was impossible in the past, “but this barrier has now been breached.”
In July last year, LUMICKS announced a collaboration with the Dutch biotech Glycostem to improve the screening of NK cells and make it easier to select the best cells for developing into cell therapies.
Overall, NK cell therapies display an increasing enthusiasm from researchers and investors, which has translated into significant investments. Going forward, one of the main directions of companies in the field is to expand NK cell therapies to more tumor types.
“In the last few years, basic biology, in vivo data, and further clinical experience shows the ability of NK cells to home to tumors, harbor memory-like features and persist in patients for much longer than expected. These intriguing data open up an untapped dimension of NK cells,” Holm noted.
“The likelihood that NK cells will be used as allogeneic, off-the-shelf products is currently bigger than it is for T cells, or at least they are catching up rapidly,” Reijmers added.
This article was updated on 05/03/2021 to correct the headquarters location of Anthony Nolan. This is an updated version of an article published on 30/10/2020.
Cover image from Elena Resko. Body text image from ONK Therapeutics. Embedded data visualizations from Jon Smith.





