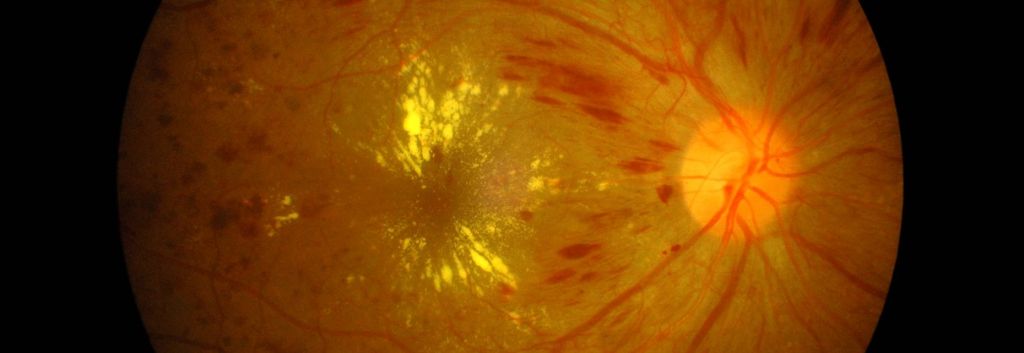
Oculis has increased its series B fundraising round to CHF35M (€31M) to continue development of its pipeline of topical eye treatments for diseases such as diabetic macular edema.
The company specializes in creating special nanoparticle formulations of drugs for treatment of various eye diseases to allow them to be applied directly to the eye as a topical treatment, rather than given by injections or implants. Its lead candidate is currently being tested at Phase II for treatment of diabetic macular edema.
Oculis began as a startup in Iceland, but is undergoing a period of expansion and moved its corporate headquarters to Lausanne in Switzerland last year.
“Our strategy is to build a global specialty ophthalmology company and as such, Switzerland represents a great foot print for us in Europe before expanding in other continents,” Oculis CEO, Riad Sherif, who also joined the company last year, told me.
“We believe Oculis to be very well positioned to build and advance a portfolio of innovative therapies for eye diseases targeting significant medical unmet needs.”
In addition to advancing its existing drug candidates, the Icelandic-Swiss biotech also plans to use the extra CHF15M (€13.3M) funding to expand its portfolio of treatments. For example, it has licensed a topical anti-TNF alpha antibody from Novartis this month, which has been tested in three Phase I clinical trials. It shows promise for treating inflammatory eye diseases affecting the anterior segment of the eye, such as dry eye disease.
Sherif said the antibody is a “perfect fit” for the Oculis’ pipeline. “It is already in a topical formulation and has shown very promising results in three clinical trials including controlled studies under IND.”
Sherif says that his focus for 2019 is to get the company’s two lead drug candidates closer to the clinic and to look for new indications for its portfolio. Longer term he has ambitions for the company to become a global player in ophthalmology.
Images via Shutterstock