The first outbreaks of Ebola (also known as Ebola haemorrhagic fever) have occurred in isolated villages in central Africa, near tropical forests, but the recent outbreak in West Africa has affected major urban centers as well as rural areas. The Ebola virus disease shows an average fatality rate about 50%. In previous outbreaks, the rate went from 20% to 90%. The virus is transmitted to humans from wild animals and then spreads in populations by human transmission. Early care support focused on rehydration and symptomatic treatment improves survival rates. No approved treatment has yet demonstrated its ability to neutralize the virus, but several treatments (immunotherapies or drug) are under consideration. There is currently no vaccine approved against Ebola, but several candidates are being evaluated. The outbreak of Ebola Guinea which started late December 2013 has 4,922 deaths recorded among the 13,703 cases, according to the latest WHO report dated October 29th, 2014. Ebola is leading to one of major health crisis of the 21st century, which is mentioned in all major media around the World. Lot of true but also lot of false. But what is really the True Story about Ebola?
History and Discovery
It all starts in Antwerp, in September 1976, with a blue Thermos. Carried in hand luggage by a passenger on a flight of the Belgian airline Sabena from Kinshasa, the capital of Zaire (former Belgian Congo). The bottle is made in the microbiology laboratory of the Institute of tropical Medicine in Antwerp.
One of the people who take delivery is a 27 years old doctor, Peter Piot. Student, he did not listen to the advice of his teachers that were saying that there was no future in the infection diseases area.
“In the Thermos, dabbling in half melted ice were two glass tubes, one of which was broken, luckily the second tube was intact.” Recalls Peter Piot, now director of the prestigious London School of Hygiene & Tropical Medicine.
A handwritten note of a Belgian doctor working in Zaire accompanies the samples. The tubes, teaches Peter Piot, contain the blood of a dead sister missionary, for which he suspected cases of yellow fever. Microscopic examination of the cells revealed a giant virus, like a worm with a similarity of Marburg virus, causing a haemorrhagic fever as well.
In Zaire, meanwhile, the outbreak is growing, with a high fatality rate. “We received a telegram from the World Health Organization (W.H.O.) requiring us to stop all research on this virus, considered very dangerous. But we had already sent a sample to the US Centers for Disease Control based in Atlanta, who taught us that it was not the Marburg virus.”
An international health mission was sent to Zaire to have a closer look on this extending outbreak in Kinshasa, which, at that time, had 3,000,000 inhabitants. This was an important government policy issue. It consisted of Americans, a French – Pierre Sureau from Pasteur Institute who was an expert for the W.H.O.- a South African and Peter Piot. The latter was the youngest because many people more experienced refused the mission.
The team collected blood samples of patients to confirm that this virus was the same that observed in Antwerp. Doctors also investigated its mode of transmission. Is it from person to person? Or by a vector? A mosquito? Another animal? The people of Yambuku (North Zaire) and surrounding villages were interviewed. How is the evolution of this outbreak? Where were the patients when they were infected? Peter Piot and colleagues first notice that the further away from the hospital, the less cases they found.
The vast majority of patients are adults. This goes against the hypothesis of an infection transmitted by a mosquito, which obviously would not save the children.
In addition to the lack of hygiene in hospitals, the team quickly discovered another mode of transmission. Many infected people attended a funeral the week before the symptoms appeared. They participated in the funeral rites, including washing the corpse, often stained vomiting, diarrhoea or blood from haemorrhage due to illness. Highly contaminating secretions.
Thus, at the hospital in this town, the first case of Ebola haemorrhagic fever was identified in September 1976 by Peter Piot, Pierre Sureau, and the members of this operation, announcing a first outbreak that hit 318 people killing 280 among them (88%). Analysis of samples will confirm that it was a previously unknown virus. The Ebola virus is named in reference to a river passing near the town of Yambuku.
Peter Piot returned to Yambuku in February 2014, on the occasion of his 65th birthday. He found there, Sukato Mandzomba, who worked as a nurse at Mission Hospital in 1976 and had been infected with the virus. “Not only Sukato Yambuku still lives in, but now it is he who ably leads the laboratory of the hospital,” said Peter Piot.
Characteristics of the virus
Phylogenetic characteristics
Ebola virus is one of at least 30 known viruses capable of causing viral haemorrhagic fever syndrome.
All of the viruses that cause viral haemorrhagic fever syndrome identified to date are RNA viruses with a lipid envelope, all are considered zoonoses, all damage the microvasculature (resulting in increased vascular permeability), and all are members of one of the following 4 families:
• Arenaviridae
• Bunyaviridae
• Flaviviridae
• Filoviridae
The known members of the family Filoviridae are Ebolavirus and Marburgvirus. According the 2012 virus taxonomy of the International Committee on Taxonomy of Viruses, Ebolavirus is classified into the following 5 separate species:
1. Sudan ebolavirus
2. Zaire ebolavirus (Origin of the 2014 outbreak)
3. Tai Forest ebolavirus
4. Reston ebolavirus
5. Bundibugyo ebolavirus
Structural characteristics
Filoviruses such as Ebola virus share a characteristic filamentous form, with a uniform diameter of approximately 80 nm but a highly variable length. Filaments may be straight, but they are often folded on themselves (Fig. 1).
Ebola virus has a non-segmented negative-stranded RNA genome containing 7 structural and regulatory genes. The Ebola genome codes for 4 structural proteins (VP30, VP35, nucleoprotein, and a polymerase protein [L]) and 3 membrane-associated proteins (VP40, glycoprotein [GP], and VP24).
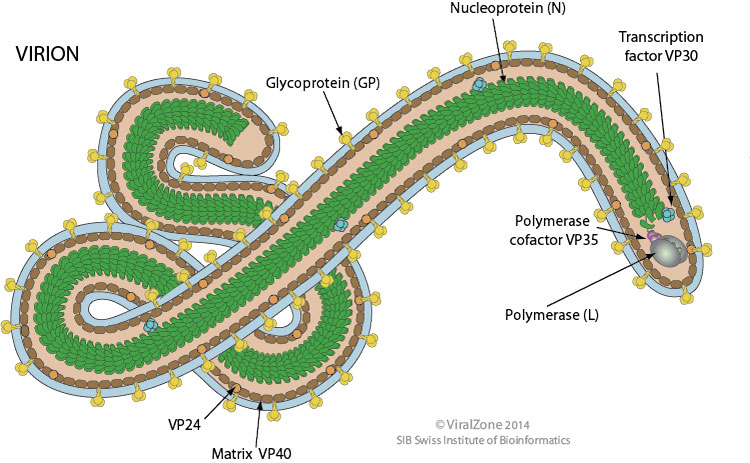
Figure 1. Structure of the Ebola virion
Most filovirus proteins are encoded in single reading frames; the surface GP is encoded in 2 frames (open reading frame [ORF] I and II). The ORF I (amino-terminal) of the gene encodes for a small (50-70 kD), soluble, non-structural secretory glycoprotein (sGP) that is produced in large quantities, early in the Ebola infection.
The sGP binds to neutrophil CD16b, a neutrophil-specific Fcγ receptor III, and inhibits early neutrophil activation. The sGP also may be responsible for the profound lymphopenia that characterizes Ebola infection. Thus, sGP is believed to play pivotal roles in the ability of Ebola to prevent an early and effective host immune response. One hypothesis is that the lack of sGP production by Marburg virus may explain why this agent is less virulent than African-derived Ebola virus. A second, larger (120-150 kD) GP, transmembrane glycoprotein, is incorporated into the Ebola virion and binds to endothelial cells but not to neutrophils. Ebola virus is known to invade, replicate in, and destroy endothelial cells. Destruction of endothelial surfaces contributes to the haemorrhagic manifestations that characterize many Ebola infections.
Transmission and Mechanisms of dispersion
African-derived filovirus infections are characterized by transmission from a host to humans or nonhuman primates, presumably via direct contact with body fluids such as, but not limited to saliva or blood or other infected tissues. Evidence in nonhuman primates indicates that Sudan ebolavirus and Zaire ebolavirus may be transmitted by contact with mucous membranes, conjunctiva, pharyngeal and gastrointestinal surfaces; through small breaks in the skin; and, at least experimentally, by aerosol. It is spread between human by direct contacts (injured skin or mucous membranes) with blood, secretions, organs or body fluids of infected people or surfaces and materials (eg. bed linen, clothing) that have been contaminated. Affected individuals are contagious from the onset of symptoms (fever) and remain contagious as long as the virus is present in their blood and body fluids, including semen and breast milk. Sperm can continue to transmit the virus up to seven weeks after clinical recovery.
In 1996, members of the National Institute for Virology of South Africa evaluated the infectivity of Ebola virus for 24 species of plants and 19 species of vertebrates and invertebrates and thus has been identified a likely reservoir for filoviruses. Insectivorous and fruit bats (Pteropodidae family) were found to support Ebola virus replication without dying. Dogs have been shown to acquire asymptomatic Ebola infections, possibly by contact with virus-laden droplets of urine, faeces, or blood of unknown hosts.
Human infection with African-derived strains has often occurred in caregivers (either family or medical) and in family members who have prepared dead relatives for burial. Late stages of Ebola virus disease are associated with the presence of large numbers of virions in body fluids, tissues, and, especially, skin. Individuals who are exposed to patients infected with Ebola without proper barrier protection are at high risk of becoming infected. During the first recorded outbreak occurred in 1976, in Yambuku, where 316 patients were infected, admission to the hospital greatly amplified the frequency of transmission. The lack of proper barrier protection and the use and reuse of contaminated medical equipment, especially needles and syringes, resulted in rapid nosocomial spread of infection. Only after adequate barrier protection and alteration in burial rituals were implemented was the outbreak contained. Health workers were often infected by treating suspected or confirmed cases of Ebola virus disease. This occurred through close contact with patients, when simple precautions were not enforced. Funeral rites in which parents and friends of the deceased are in direct contact with the body can also play a role in the transmission of the Ebola virus.
Ebola virus infection has no sexual predilection, but the infected population, men and women differ in which direct exposure occurs. Men, by the nature of their work exposure in forest and savanna regions, may be at increased risk of acquiring a primary infection from gathering “bush meat” for food, as well as an unknown vectors. Because women provide much of the direct care for ill family members and are involved in the preparation of the bodies of the deceased, they may be at increased risk of infection. However, men and women who are medical healthcare providers seem to share a high and equal risk of infection.
Symptoms and Diagnosis
The incubation period, meaning the time between the infection and the onset of symptoms, ranges from 2 to 21 days. As long as they do not show symptoms, humans are not contagious. First symptoms are a sudden, fever fatigue, muscle pain, headache and sore throat. They are followed by vomiting, diarrhoea, rash, symptoms of kidney and liver failure and, in some cases, internal and external bleeding (eg. bleeding gums, blood in the stools). Laboratory tests revealed lower white blood cells and platelets counts but elevated liver enzymes.
The Ebola virus disease can be difficult to distinguish from other infectious diseases such as malaria, typhoid fever and meningitis. The following investigations are used to confirm the infection by Ebola :
• immunoassay (ELISA)
• antigen capture assay
• seroneutralisation assay
• reverse transcription followed by a polymerase chain reaction (RT-PCR)
• electronic microscopy
• virus isolation on cell culture
Treatments
Currently, no specific therapy is available that has demonstrated efficacy in the treatment of Ebola haemorrhagic fever, and there is no commercially available Ebola virus vaccine.
Rehydration orally or intravenously and treatment of specific symptoms improve survival rates. In these conditions, supportive therapy with attention to intravascular volume, electrolytes, nutrition, and comfort care is of benefit to the patient. Intravascular volume repletion is one of the most important supportive measures.
Survivors can produce infectious virions for prolonged periods. Therefore, strict barrier isolation in a private room away from any traffic must be maintained throughout the illness. Patient’s urine, stool, sputum, and blood should be properly destroyed and any materials that have come in contact with the patient or the patient’s body fluids, if they are not single-use only, disinfected with a 0.5% sodium hypochlorite solution. Patients who have died of Ebola virus disease should be buried promptly and with as little contact as possible.
In those patients who do recover, recovery often requires months, and delays may be expected before full resumption of normal activities. Weight gain and return of strength are slow. Ebola virus continues to be present for many weeks after resolution of the clinical illness. Therefore, it is important to maintain this quarantine several weeks.
Experimental treatments
At present, no specific anti-Ebolavirus agents are available. Agents that have been studied for the treatment or prevention of Ebola virus disease include the following:
• Ribavirin (possesses no demonstrable anti-Ebolavirus activity in vitro and has failed to protect Ebolavirus -infected primates)
• Nucleoside analogue inhibitors of S-adenosylhomocysteine hydrolase (SAH)
• Interferon beta
• Horse- or goat-derived immune globulins
• Human-derived convalescent immune globulin preparations
• Recombinant human interferon alfa-2
• Recombinant human monoclonal antibody against the envelope glycoprotein (GP) of Ebola virus
• DNA vaccines expressing either envelope GP or nucleocapsid protein (NP) genes of Ebola virus
• Activated protein C
• Recombinant inhibitor of factor VIIa/tissue factor
In 2014, an experimental treatment given to two Americans working for NGOs and infected with Ebola virus has been effective in treating the symptoms of the disease. The treatment called Zmapp has been developed by the biotechnology company named Mapp Biopharmaceutical (California, USA). The immune system of humans responds only moderately to Ebola. Also, Zmapp is made from three monoclonal antibodies (mAbs) directed against the Ebola Zaire virus strain. Humanized and replicated in the laboratory, the mAbs produced by the murine immune systems are injected in humans. The mAbs, found in their mouse form in the Zmab (Canada), were licenced from Defyrus (Toronto, Canada) and manufactured in tobacco plants in the USA. Designated at the time under the code name “MB-003”, the cocktail protected all primates to whom it was administered one hour after infection. The protection rate was still two-thirds of the animals when the MB-003 was injected 48 hours after intravenous infection. A study published in 2013 in Science Translational Medicine confirmed these results and showed that 43% of primates were still protected by an injection of MB-003 in the 104 to 120 hours after contact with the Ebola virus.
In 2014 Dr. Kent Brantly infected with Ebola, initially requested that his colleague Nancy Writebol also infected receive the Zmapp injection first, thinking himself better able to resist despite nine days post-infection. But the state of the doctor abruptly worsened, particularly respiratory level, and it was him who first received the intravenous treatment. Within an hour, his condition improved considerably, reports CNN. “The next morning, he was able to take a shower himself before boarding the plane back to Atlanta”. He has been treated in the specialized infectious diseases unit, Kent Brantly is now able to walk again. He also had been transfused with a blood bag of a 14 years old boy who survived to Ebola and thus naturally produces antibodies against this infectious agent. Nancy Writebol also received a dose, but the observed effect was less. However, a second dose resulted in a “significant improvement in her condition” notes CNN, which allowed her to be repatriated to the United States. The role of this treatment in the health status of these two Americans is not yet known, the mortality rate of the virus ranging from 20 to 90%.
In France, four experimental treatments are allowed since September 18th 2014: The TKM-100-802 (Canada), the Zmab (Canada) close to the Zmapp (USA) also authorized but currently unavailable, and the Favipiravir (Japan).
TKM-100-802 (Tekmira, Canada) : RNAi therapeutic utilizing Tekmira’s Lipid Nanoparticle. This technology represents the most widely adopted RNAi delivery technology to date. These RNAi blocs virus synthesis within cells infected by Ebola Virus.
Zmab (Public health agency, Winnipeg, Canada): is a cocktail of three mouse mAbs: m1H3, m2G4 and m4G7 developed at the National Microbiology Laboratory of the Public Health Agency of Canada in Winnipeg.
Favipiravir (Toyama Chemical, Japon) : also known as T-705 or Avigan, is an experimental anti-viral drug with activity against many RNA viruses. It is a pyrazinecarboxamide derivative. The mechanism of its actions is thought to be related to the selective inhibition of viral RNA-dependent RNA polymerase.
Future therapies
A new study reveals that a protein of the Ebola virus can transform into three distinct shapes, each with a separate function that is critical to the virus’s survival. Each shape offers a potential target for developing drugs against Ebola virus.
Erica Ollmann Saphire’s team of The Scripps Research Institute analyzed the structure of VP40, a protein best known for its role in creating and releasing new copies of the virus from infected cells. “The interesting thing about VP40 is that it does more than that, we found that it is multifunctional, with several essential roles for the virus” said Erica Ollmann Saphire. The team discovered that the protein can alter its shape, causing multiple copies of the protein to join up and create three very different assemblies: a butterfly shape composed of two, a ring formed by eight, and a linear structure built from six VP40 molecules. Prior to the study, only the protein ring was known.
The function of each structure is linked to a specific stage of the virus life cycle. While moving around inside infected cells, VP40 assumes the butterfly shape. In the early stages of an infection, the VP40 molecules change their structure and assemble into a ring near the cell nucleus, regulating how the virus’s genetic information is copied. In the later stages, VP40 travels to the cell’s outer layer, or membrane, and transforms into its linear structure, which plays a crucial role in the creation of new copies of the virus. All three functions of VP40 – traveling inside infected cells, regulating genetic information and creating new viruses – are essential to the Ebola virus, and disrupting any of the corresponding structures or their transformations would severely affect it. Therefore, VP40’s triple role provides researchers with important clues for the development of potential antiviral drugs. Although a cure for Ebola virus disease is still not available, the new study already has practical applications: The same VP40 proteins produced for this study are being used in test strips to identify the disease in patients affected by the current outbreak in West Africa.
Vaccines
Canada / USA : The vaccine has been developped by the Canadian public health agency based in Winnipeg and NewLink Genetics who own the commercialisation licence for the “rVSV-ZEBOV”. rVSV-ZEBOV uses the attenuated virus of cattle disease on which has been grafted an Ebola virus gene.
Great Britain : The vaccine “cAd3-ZEBOV” developped by GlaxoSmithKline (GSK) use an adenovirus derivated from the chimpanzee as a vector on which has been inserted a gene fragment of Ebola virus.
France : While the strategy of France’s research will be presented shortly, a team based in Lyon at the International Centre for Infectious Diseases Research (Ciri), who has already developed the bases of two experimental vaccines, fears of not receiving the funds to develop a new candidate vaccine. “We believe this new vaccine can be best in terms of protection and safety” says Viktor Volchkov, the team leader, which notes that the rVSV-ZEBOV vaccine candidate – born of a Franco-Canadian collaboration – pose “an ethical problem” because it comes from a not existing in nature viruses. The new French vaccine should also be “easy to produce in large quantities” thinks Volchkov, contrary to the Russian and cAd3-ZEBOV vaccines. The cAd3-ZEBOV would also have the disadvantage of having to be administered at high doses to be effective. French candidate vaccine “may be ready for testing in a few months” says Volchkov. Virologist redoubt, as it was the case the two previous times, not to receive the funds necessary to carry out these tests. “The government thinks only of the present events when he has to think for the future” laments Volchkov while the pharmaceutical industry only contacts the researcher without moving forward. We would have the uncomfortable feeling to think that in France, it would be pointless to get into a race where others are now ahead of us.
Russia : While the virus has killed more than 4,000 people, the Russian Minister of Health, Veronika Skvortsova, said that Moscow could provide three vaccines within six months. “One is ready for clinical trial” she said, adding that one of the vaccines had been created from an inactive strain of the virus. Russia, which has not reported any cases of infection on its territory, sent late August a team of virologists and a mobile laboratory to help fight against the outbreak in Guinea. The minister said 71 Russian airports are equipped with thermal cameras to detect the first signs of infection. “Basically, [the virus] comes from West Africa. Consequently, all students from West Africa in Russian universities, and it is more than 450 people are under constant surveillance” she added.
Several preliminary trials on the way
W.H.O. and partners have worked together to achieve as soon as possible early clinical trials in healthy volunteers, known as Phase 1 trials, in order to provide raw data on the safety of vaccine candidates and their ability to induce an immune reaction in humans. Several international Phase 1 trials are underway and others should start shortly in a dozen sites in Africa, Europe and North America. After encouraging results in the trials of cAd3-ZEBOV in monkeys, a preliminary study phase in humans was launched early October on healthy volunteers in the United States and the United Kingdom. It will also soon be held on the African continent, Mali and Ghana. A total of 160 volunteers will participate. A Phase 1 with the rVSV-ZEBOV started early October in the United States. Alongside the development of the industrial manufacturing process, these so-called test “Phase 1” is intended to evaluate the safety and tolerance of the vaccine candidate and a first estimation of its ability to induce an immune reaction. Doses to meet the international quality standards for Phase 2 vaccines are expected between 2015 and 2016. It is only after the results of these Phase 2 trials will be obtained, that a large-scale vaccination may be planned (Phase 3) to strengthen the knowledge around dosage and induced protection.
Dr. Ripley Ballou, program director of Ebola vaccine research at GSK, said on Oct. 17th, that the complete data security and efficiency would not be available before the end of 2015. GSK hopes to have 20,000 doses ready for testing early next year. “We must be able to manufacture the doses according to the use in clinical practice and it will take us far in 2016 to reach this point. I do not think we can consider it as the first response to this current outbreak, but on-going trials will help in the future” said Dr. Ballou.
“Hearing GSK assert that Ebola vaccine will arrive too late is disappointing. We acknowledge and endorse the highly accelerated GSK vaccine development, but efforts must be intensified, as we believe that a vaccine would be important for this outbreak, as well as prevent and control Ebola in the future. Nobody knows how long this outbreak will last. Our patients, those on the front lines and the people of West Africa can not hear that it’s too late” responded Dr. Manica Balasegaram of the NGO named Doctors Without Borders.
Accelerate the development and manufacture of a vaccine is essential, but it is not about doing anything, everyone agrees on that. In this race against the clock, Dr. Ballou (GSK) reveals procrastination when discussing the development of a vaccine against Ebola with W.H.O. in March 2014, while the outbreak had already started. This need was, it seems, discarded. “No one anticipated that we would need a vaccine. Thus, internally and – I believe – the W.H.O., thought that the best approach would be to closely monitor the outbreak”. Seven months later, with a virus out of control, Dr. Ballou concedes “In retrospect, I think we should have started the action sooner. But, you know, what’s done is done, and we work closely with the W.H.O. There is no need to point at one or the other about it”.
Drawing lessons from vaccine trials in Africa against malaria, HIV, cholera, meningitis, hepatitis B, experts are urging the international community to never invoke anymore the “market failure” to justify delays in the provision of vaccines against Ebola and other diseases.
Key numbers dated 13th October 2014
W.H.O. calls this outbreak “Worldwide public health emergency” and believes that it could lead to more than 20,000 cases of contamination by November and 10,000 cases per week by December 2014. Ebola haemorrhagic outbreak starting from Guinea end of December 2013 (a survey of the New England Journal of Medicine found the patient “Zero” in the person of a young boy living in a rural area of south-eastern Guinea) already killed 4,922 people among the 13,703 cases recorded according to the latest report published by the W.H.O., in October 29th.
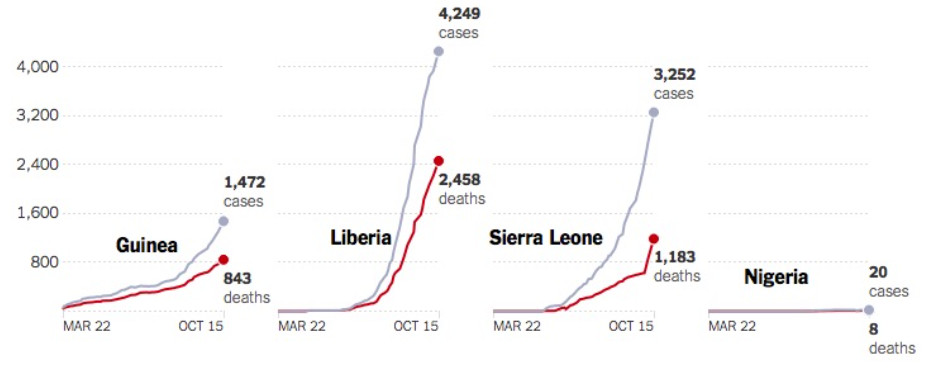
Liberia is today the most affected country, with nearly 6,000 cases and “only” two thousand confirmed (meaning that another 4,000 are awaiting confirmation, so recent). Sierra Leone, with 2,593 confirmed cases and 753 confirmed deaths, struggles to stabilize the situation. The outbreak has also flared up in Guinea, where it was born, after a lull during the summer (Fig. 2). Nigeria and Senegal however, seems containing the disease or even better for Nigeria that seems to be now free of Ebola as a recent W.H.O. announcement. Indeed, Forty-two days, twice the maximum incubation period of the virus (21 days) have passed with no new cases in Nigeria. “Nigeria is now rid of Ebola” said Rui Gama Vaz, W.H.O., at a press conference in Abuja (Nigeria).
More than 400 health care workers in West Africa have been infected with Ebola during the current outbreak, and 233 had died by October 8th. The W.H.O. said that the high rates of infection among medical workers could be attributed to shortages or improper use of protective equipment; not enough medical personnel; and long working hours in isolation wards.
At least 17 cases have been treated in Europe and the United States (Fig. 3). Most involve health and aid workers (Tbl. 1) who contracted Ebola in West Africa and were transported back to their home country for treatment. A Spanish nurse who was the first person known to have contracted the Ebola virus outside Africa was declared free of the disease recently after being treated by Zmapp.
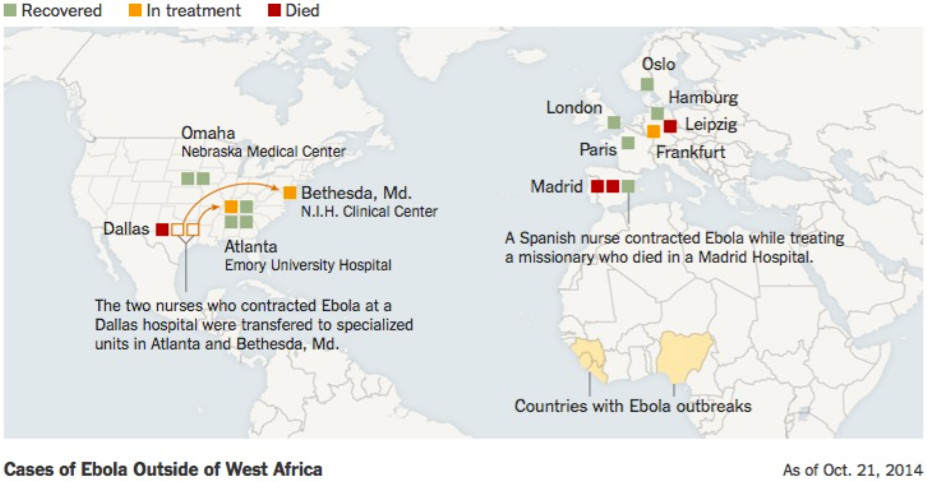
Figure 3. Map of Ebola outbreak in 2014.
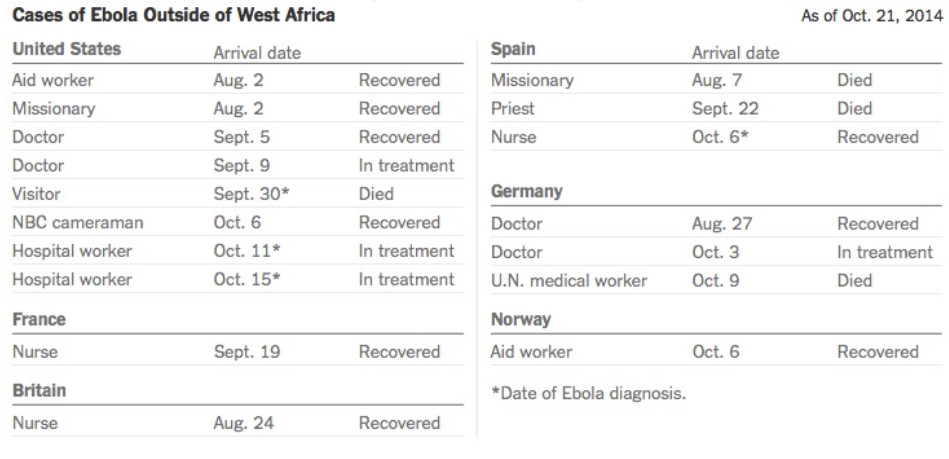
Table 1. Cases of Ebola patients outside of Africa during the 2014 outbreak (October 21st,2014)
Bioterrorism: Ebola is not a biological weapon transforming the world into “Zombie-land”
The virus is not, as could have been claimed by Forbes and The Daily Mail, a blunt instrument that the organization of the Islamic State could incorporate into its arsenal of bioterrorism. It is not the last resort of a lonely wolf on a suicide mission, as the words of Fox News. And it is not, as stated by the leader of the Nation of Islam, a biological weapon of racial profiling crafted by the United States as proved by the case of Western patients infected.
Ebola is perfectly real and frighteningly dangerous. It is therefore essential to support research to eradicate this scourge and protect the people, all vulnerable as diverse as they are.
First, the virus is not a good candidate to become a biological weapon. It does not spread quickly – its factor R0, or the measure of the infectiousness of the virus, is pretty much of 2. This means that in a population where everyone is at risk, every infected person will infect, an average of two others. Given that an individual carrying Ebola is contagious only when symptoms manifest. Thus we are likely to contain an outbreak in countries with an efficient system of public health.
Unlike existing biological weapons such as anthrax (Anthrax), the mode of transmission of Ebola virus is very difficult to be converted into a weapon. Anthrax spores can be dried and pulverized to make particles streaming in the air that could be inhaled. To be passed, Ebola requires bodily fluids and, if we try to make weapons, they are no vectors neither very reliable nor very effective. The study of E. JOHNSON in 1995, expressing the possibility of nebulized Ebola, taking the form of aerosols for the successful contamination of non-human primates applies in a limited framework for high technology and strict conditions of temperature, hygrometry and virus concentration.
Certainly, filoviruses have infected humans with no known history of parenteral or ocular inoculation of the virus and a respiratory route of infection cannot be ruled out. Those cases may have resulted from exposures to unusual concentrations of aerosolized virus not often encountered naturally.
As we saw, the virus do not spread easily from person to person during the Ebola outbreaks in Africa, and rates were highest in individuals who were in direct physical contact with a primary case. The rates were 3.5 times higher in people who provided nursing care than in those who were in casual contact with a primary case; no cases occurred in children whose only known exposure to the virus was sleeping in the huts occupied by their fatally ill parents. Although coughing was common among the human Ebola haemorrhagic fever cases in Africa, there was no direct evidence for aerogenic spread of Ebola virus in human populations.
Conclusion
Ebola, this viral disease recently discovered, first named in 1976 and that we now know the high lethal potential, had made only a small number of deaths so far. With this new outbreak, in addition to previous ones, Ebola is now involved in nearly 15,200 cases including more than 6,000 deaths. This single outbreak in 2014 is responsible for 4,922 deaths among the 13,703 cases recorded according to the latest report published by the World Health Organization, October 29th 2014.
Different treatments in their experimental stages, are used in the Western world but their success remains uncertain.
Meanwhile several vaccines are in development whether in America, Canada, Europe and Russia with results expected soon. The question on everyone’s mind is: will they be ready on time and in what volumes if the oubreak spreads?
We can notice the large use of biotechnology or immuno-biotechnology in the experimental treatments implemented.
At present, vaccines are emerging and are part of a prevention approach that complements the therapeutic arsenal also produced through biotechnology.





