Sensorion is about to start Phase II trials to treat acute severe vertigo. What makes the inner ear such an attractive target for biotech?
Sensorion is a French biotech developing treatments for inner ear pathologies, including vertigo, tinnitus and hearing loss. The company has just announced the start of Phase II trials with SENS-111, a drug candidate to treat acute severe vertigo.
Also known as acute unilateral vestibulopathy, acute vertigo appears suddenly and causes intense nausea and vomiting. These severely debilitating symptoms require hospitalization for a couple of days and rehabilitation of the patients.
Sensorion’s candidate is an oral neuromodulatory drug targeting the histamine type 4 receptor. It was originally developed by the Spanish Palau Pharma, which gave Sensorion an exclusive license to develop inner ear treatments.
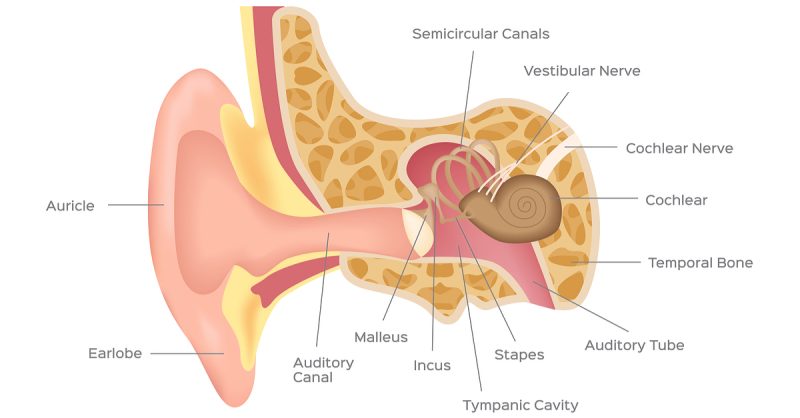
The interest in the inner ear pathologies market, estimated at over €10B, stems from the lack of effective and well-tolerated drugs in this area. However, Sensorion is not the only one to have laid eyes on this niche: AudioCure Pharma, from Germany, just raised €9M to start Phase I trials for a treatment dealing with acute hearing loss and tinnitus. Auris Medical, in Switzerland, is leading the pack with two candidates in Phase III to treat tinnitus and sudden deafness.
Other companies are taking a whole new approach: Germany-based Sonormed is developing a prescription app, Tinnitracks, aimed at reducing the effects of tinnitus using the patient’s favorite music. Whoever takes the lead, all these exciting developments are giving hope to the 140 million people suffering from inner ear conditions worldwide.
Featured image by Darren Baker/shutterstock.con; figure by Gritsalak karalak/shutterstock.com





