Norwegian company SoftOx Solutions AS says it has received the final report and analysis of its phase 1 study evaluating the safety and tolerability of SoftOx Biofilm Eradicator (SBE-01).
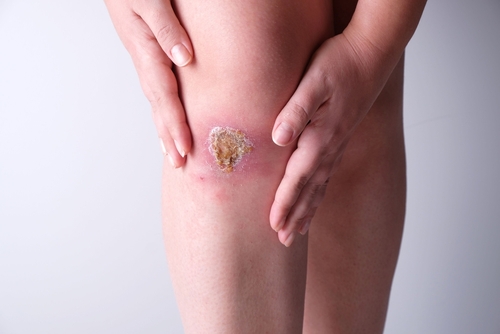
SBE-01 is SoftOx’s antimicrobial solution for the treatment of chronic wounds.
The study is a first-in-human investigation of the safety, tolerability, and antimicrobial efficacy of single and multiple doses of SBE in patients with chronic leg wounds. The phase 1 study was divided into two parts: a randomized, blinded, single ascending dose (SAD) phase, followed by an open label, multiple ascending dose (MAD) phase, where patients were treated once-daily or twice-daily over five days.
Key findings about SoftOx’s SBE-01
The final report reaffirms previous findings that SBE is safe and well-tolerated by patients suffering from chronic leg wounds. There were no statistically significant differences in the evaluation of pain during and after the wound cleaning procedure comparing SBE with placebo. The study found that formulations of SBE consistently reduced the bacterial burden in the wound compared with pre-treatment. There was also evidence of wound healing with a reduction in wound area of between 24 to 36% after just 5 days’ treatment.
Thomas Bjarnsholt, chief scientific officer at SoftOx, said: “According to the World Health Organization, antimicrobial resistance is one of the largest threats to human health, which is why it is critical to develop new and innovative ways to tackle bacterial infections.
“These results clearly suggest that the SBE solution is safe and well-tolerated by patients and can significantly reduce the overall bacterial burden in wounds, ultimately promoting wound healing. Taken together with previous clinical studies, these data are early proof-of-concept for the technology as a potential treatment candidate for patients suffering from chronic wounds worldwide.”
Next steps for SBE-01
Having demonstrated early proof-of-concept for the SBE project and in accordance with the company’s corporate strategy, SoftOx will seek a strategic partner to further develop and advance its wound care segment within acute and chronic indications.