Newsletter Signup - Under Article / In Page
"*" indicates required fields
The tough regulations that apply to genetically modified organisms may no longer affect plants and animals created using CRISPR gene editing technology.
The European Court of Justice has ruled that crops and drugs developed using precise gene-editing techniques like CRISPR/Cas9 may not need to be regulated as strictly as genetically modified organisms (GMOs). So far, uncertainty surrounding the existing rules has slowed the use of CRISPR gene editing technology but the European Court’s advocate general has come out in support of using these genome editing tools. The use of these approaches could help to create more resistant crops and better medical treatments for genetic diseases.
Back in 2001, a directive stated that the deliberate release of GMOs that received whole genes or pieces of DNA from other species was against EU law. However, the directive allowed mutagenesis techniques like irradiation, and confusion grew. Many breeders felt that gene editing should also be permitted as only single nucleotides are altered, while opponents believe that deliberate changes to a gene give rise to products that must be regulated in the same way as other GMOs.
Researchers like Ralf Wilhelm at the Julius Kühn Institute have welcomed the clarification of the GMO directive that was set almost 20 years ago: “This opinion goes in a clarifying direction… [The opinion] doesn’t say that all products created with the new gene-editing techniques should automatically be considered under all the obligations of the GMO regulations.” The Court will rule on this subject later in the year, and we will see if it follows the advice of its advocate general.
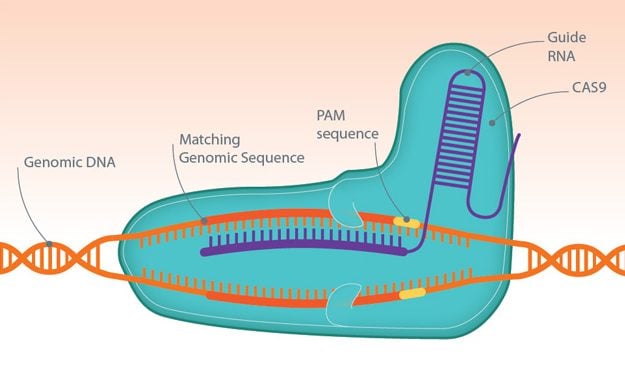
CRISPR, which stands for clustered regularly interspaced short palindromic repeats, is used in combination with Cas9 nucleases for genome engineering, by tweaking one or more target genes at a time. Short pieces of ‘guide RNA’ tell Cas9 where to break a DNA strand in order to knock out a target gene. This can be replaced with a new gene to improve the characteristics of a crop or restore the normal function of a faulty gene. In exciting news, CRISPR Therapeutics will begin the first European clinical trial using CRISPR/Cas9 technology in humans this year.
There are two major concerns if gene-edited products are subjected to the GMO’s strict directive. First, increased checks and controls would make it even more expensive for European academics and small companies to develop products for the market. Second, the public could reject these products if they are considered GMOs, as was the case for genetically-modified foods. CRISPR gene editing technology could play a key role in the fight against new diseases that arrive in Europe due to globalization and climate change, so the prospect of slowing their progress even further is worrying.
What’s more, it appears that there is nothing stopping individual EU member states from making their own rules for gene editing. This raises the possibility that there could be a patchwork of different rules across the continent, which could undermine the single market.
Images – Gts / shutterstock.com; unoL / shutterstock.com






