Newsletter Signup - Under Article / In Page
"*" indicates required fields
Boehringer Ingelheim’s fight against liver disease is taking shape, having agreed on a partnership with RNA pioneer MiNA Therapeutics to fight NASH.
British biotech, MiNA Therapeutics, has announced a partnership with pharma giant Boehringer Ingelheim to focus on developing compounds to treat fibrotic liver diseases like Non-Alcoholic SteatoHepatitis (NASH). Bringing MiNA on board has given Boehringer Ingelheim access to the biotech’s small activating RNA (saRNA) technology, meaning the big pharma will now be able to use multiple mRNA-regulation technologies. It is no wonder that Boehringer Ingelheim is ramping up its efforts, with the NASH market expected to be worth up to $35B (€30B).
Many people are hoping that a NASH treatment will arrive soon, as it is believed to affect 10% of the population and could be the main cause of liver transplants by 2020. The condition is characterized by the build up of fat in the liver due to an unbalanced diet or lack of exercise. This accumulation causes inflammation, which can cause fibrosis, potentially developing into cirrhosis.
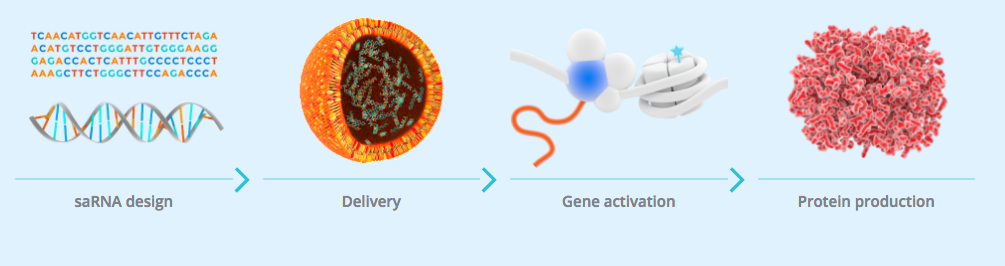
MiNA’s saRNAs are small oligonucleotides that could help us to access previously undruggable targets. The saRNAs are loaded onto Argonaute proteins in the cytosol and translocated into the nucleus. Once there, the Argonaute complex associates with a target gene and stimulates transcription by recruiting the necessary proteins. This allows the expression of proteins with therapeutic potential. The platform has already been tested in the liver, during a first-in-man study for cancer last year.
Pharma companies have been snapping up NASH-focused biotechs like nobody’s business, including Allergan’s acquisitions of Akarna and Tobira and Gilead swallowing up Nimbus. But, biotechs are becoming increasingly determined to remain independent, they might have to settle for partnerships. Boehringer Ingelheim will hope that its partnership with MiNA gives its NASH R&D division a boost.
Genfit is leading the race to become the first to release a drug into the NASH market, with its candidate, Elafibranor, undergoing a Phase III trial. Meanwhile, Allergan and Novartis have come together in an attempt take over the NASH space by combining Allergan’s anti-fibrotic candidate, CVC, with Novartis’ Farnesoid X receptor (FXR) agonist. Inventiva’s PPAR activator and the Tiziana’s monoclonal antibody are also active in the field.
Although MiNA and Boehringer Ingelheim are a little way behind in the race to bring the first NASH treatment onto the market, Genfit CSO, Dean Hum, believes that there will be room for many different approaches – so all is not lost.
Images – u3d / shutterstock.com; MiNA Therapeutics