Newsletter Signup - Under Article / In Page
"*" indicates required fields
Cell therapy is revolutionizing medicine, here’s how the latest technology can help overcome the major challenges stopping it from taking over the market.
Offering unprecedented possibilities to treat some of the most challenging diseases, cell therapy is stealing the show in the biotech space. Strimvelis, the first hematopoietic stem cell (HSC) gene therapy is already treating rare genetic diseases. In less than a year, CAR-T therapy is expected to hit the market and revolutionize the treatment of cancer. And in the not-so-far future, cell therapy could even eradicate HIV or put an end to diabetes, which is reaching epidemic proportions in Western countries.
The first cell therapies have already been on the market for a few years, and analysts are confident that the numbers will quickly grow over the years. The stem cell therapy market alone is expected to hit €57Bn ($61Bn) by 2022, and the upcoming CAR-T technology will reach an impressive €8Bn ($8.5Bn) in the next decade.
Although the potential is definitely there, researchers are still looking for ways of making these therapies cheaper, safer and more effective. Currently, not all patients are suited to receive cell therapy due to scientific or economic challenges. The advent of allogeneic therapies is addressing the financial obstacles, but what about efficiency?
Challenges in Cell Therapy
One of the key elements to building a successful cell therapy are viral vectors, which are used to deliver the necessary DNA sequences to engineer the cells. Lentiviral vectors are a common choice because they have a rather larger capacity and enable long-lasting genetic expression.
They are specially researched for ex-vivo treatment of hematopoietic stem cells and primary T-cells. The percentage of cells that can be reached, however, often remains low, and the number of gene copy numbers per cell can be extremely variable.
Finding a solution to this challenge is unfortunately not as simple as increasing the virus load. Cells that carry a surplus of copies integrated into their DNA are more prone to mutations that affect their survival and put the safety of the patient in danger. This is an essential factor taken into account during the regulatory phase to determine whether a therapy can make it to the market or not.
Boosting Efficacy
How can we then improve the efficacy of transduction without affecting the health of the cells and the safety of the therapy? In the lab, researchers often use enhancers such as polybrene, a polymer that can increase the efficacy of transduction of viral DNA. However, this substance is not applicable in a clinical context because of its heightened cell toxicity.
To overcome these challenges, scientists at SIRION Biotech got down to work and screened for compounds that could improve the fusion of the viral and cell membranes. The result was a technology with the potential to solve a major problem in the development of cell therapies, with DNA delivery reaching an impressive 80% of hematopoietic stem cells while keeping the copy numbers down to the ideal value of 3 to 5 per cell.
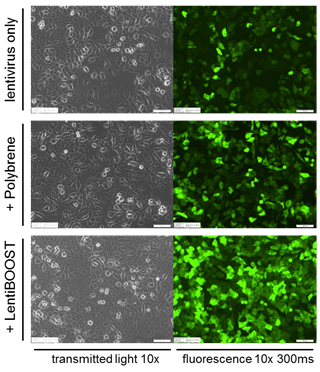
This technology, called LentiBOOST, works its magic simply by adding a non-toxic polymer to the mixture during transduction. It has already been accepted as an element of clinical trials from phase I to phase III and its possibilities seem unlimited. One of SIRION’s partners, the Heinrich Pette Institute in Hamburg, is studying the potential of LentiBOOST to improve transduction in a therapy intended to actively remove HIV from infected blood cells and induce long-lasting resistance against the virus.
With applications ranging from cancer to infectious disease, cell therapy is definitely going to change medicine. Technology like LentiBOOST is helping these amazing developments materialize with a boost to both their efficacy and safety.
You can find more information on LentiBOOST at SIRION Biotech’s website!
Images from Montri Thipsorn, vchal /Shutterstock; SIRION Biotech






