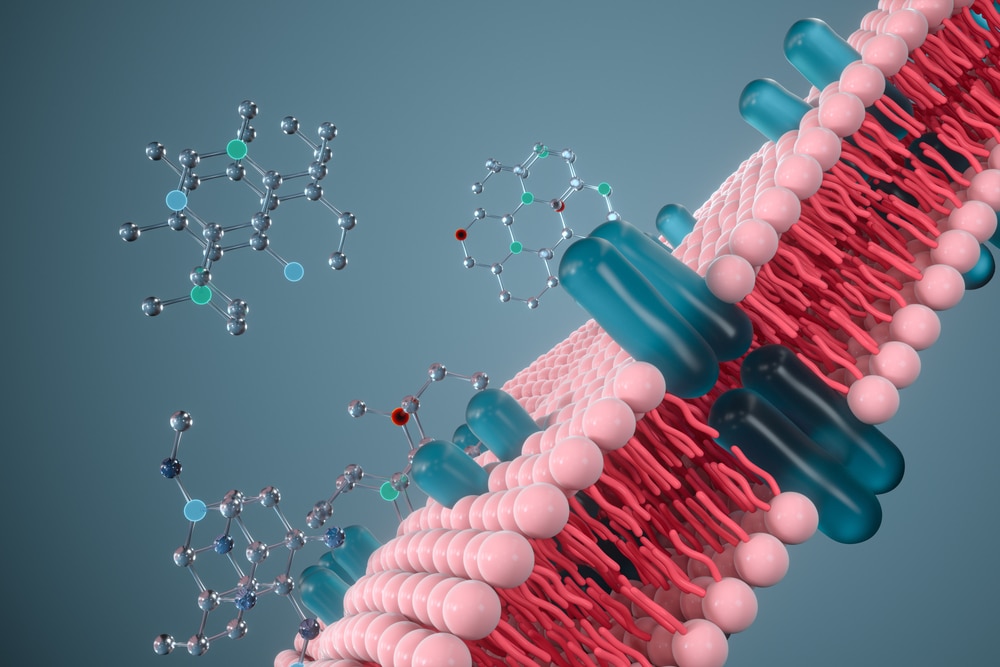
Membrane proteins are key targets for many drugs.
Some of them, called transporters, move certain substances in and out of the cellular environment. Yet, extracting and storing them for observation is particularly complex. A team from the University of Geneva (UNIGE) in Switzerland, in collaboration with the University of Zurich (UZH), has developed an innovative method to study their structure in their native environment: the cell. The technique is based on electron spin resonance spectroscopy.
The results, published in the journal Science Advances, may facilitate future development of new drugs.
In living organisms, each cell is surrounded by a cell membrane (or cytoplasmic membrane). This membrane consists of a double layer of lipids. It separates the contents of the cell from its direct environment and regulates the substances that can enter or leave the cell. The proteins attached to this membrane are called membrane proteins.
Located at the interface between the outside and inside of the cell, they carry various substances across the membrane – into or out of the cell – and play a crucial role in cell signaling, i.e. in the communication system of cells that allows them to coordinate their metabolic processes, development and organization. As a result, membrane proteins represent more than 60% of current drug targets.
Difficult to study
The biophysical study of their structure – the spatial organization of the constituent amino acids – is essential. To characterize them, scientists must extract these proteins from the cell membrane in which they are found and isolate them from all other proteins. Once extracted, membrane proteins cannot be studied in aqueous solutions. They must be maintained in liquid solutions composed of detergents. They can also be inserted into artificial membranes called nanodiscs, made of proteins and lipids, or in pure lipidic membranes.
These strategies remove them from their physiological environment and do not allow their functioning to be observed in situ. Proteins outside their native environment might show different structural properties, therefore misleading drug development.
A new method
A team led by Enrica Bordignon, full professor in the Department of Physical Chemistry at the UNIGE Faculty of Science, in collaboration with Markus A. Seeger, associate professor at the Institute for Medical Microbiology at the UZH, has developed a new method for studying membrane proteins in action in living cells; in the inner cell membranes of the intestinal bacterium E. coli. To achieve this, the research team relied on a specific tool nanobodies.
‘‘These are fragments of antibodies that are able to recognize and bind to a specific target, such as an antigen or in our case, a membrane transporter, in a very efficient way,’’ Bordignon said.
The scientists have artificially produced specific nanobodies for a membrane transporter and use them to directly report on its structure.
‘‘Inserted into E. coli cells, two nanobodies target the desired membrane protein on the inner membrane of the cell and attach to it,’’ Seeger said.
The multidisciplinary team also included scientists from the Ruhr University Bochum (cluster of excellence RESOLV) and the University of Osnabrück, Germany,and the University of Southampton, U.K.
New targets for drugs
Beforehand, a small magnetic probe (a molecule carrying unpaired electrons) was attached to each nanobody.
‘‘When two nanobodies bind to the transporter, we can measure the distance between the two magnetic probes in cells using our EPR methods,’’ Bordignon said.
This technique is called electron paramagnetic resonance spectroscopy (EPR) or electron spin resonance. The distance measured is in the nanometer range (one millionth of a millimeter).
‘‘For the first time, we have managed to obtain a clear picture of the conformation of a membrane protein in its real environment and we could follow the change induced when we modified one single amino acid into another one,’’ Bordignon added.
The new strategy allows a precise determination of membrane proteins’ properties in their direct environment. It offers the possibility of better understanding how these proteins transport certain substances into and out of the cell. This method also has the advantage of being easily transposable to mammalian cells. It could then be used to better understand and therefore better target the membrane proteins which reject certain anti-cancer drugs outside the cell, and thus combat the phenomenon of multi-drug resistance.
Partnering 2030: The Biotech Perspective 2023