Newsletter Signup - Under Article / In Page
"*" indicates required fields
German scientists say a drug already approved for other purposes can negate how tumor cells protect themselves from the immune system.
Many tumor cells mist themselves with a protective ‘perfume’ that disables the immune system. But a drug already approved for other purposes can apparently render this weapon harmless.
This is the conclusion of a German study by the University of Bonn and the University Medical Center Hamburg-Eppendorf, which has now appeared in the Journal for ImmunoTherapy of Cancer. The researchers now want to further optimize the compound. In the medium term, this could pave the way for new anti-cancer drugs.
Adenosine
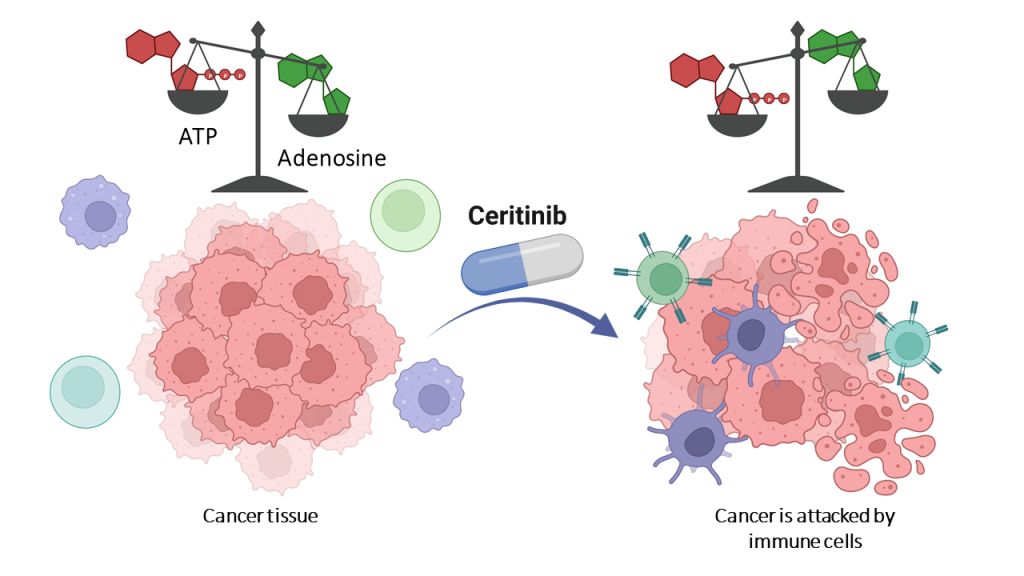
Many cancer cells surround themselves with a dense cloud of adenosine. This not only suppresses the immune system, but also it stimulates the formation of new blood vessels that supply the tumor with oxygen and nutrients. It also ensures the malignant cells migrate to other organs and form metastases there.
Adenosine is produced from adenosine triphosphate (ATP). Tumor cells secrete large amounts of it. They carry various enzymes on their surface that then convert the ATP to adenosine in several steps. One of these is known as CD39.
“It catalyzes the first of the conversion steps,” said Christa Müller from the Institute of Pharmacy at the University of Bonn.
“If CD39 is inhibited, hardly any adenosine is produced.”
Around the globe, pharmaceutical researchers are therefore searching for an active ingredient that slows down CD39. Because without adenosine, tumors would no longer be protected from the immune system.
“Instead, ATP would accumulate around the cancer cells, which would actually stimulate the immune response,” Müller said.
“So the body’s own defenses would not be suppressed; on the contrary, they would be turned on extra sharp.”
Approved active substances scrutinized
So far, the search has been largely unsuccessful. And so the University of Bonn research group took a different route.
“There are other enzymes in the body than CD39 that also process ATP,” said Laura Schäkel, Müller’s collaborator, and the person who carried out many of the central experiments in the study. “
These include, for example, the so-called protein kinases. The nice thing is that there are already approved drugs that inhibit protein kinases. We now looked at whether they also work against CD39.”
At the start of the study, there were 50 different agents approved for certain diseases that inhibit protein kinases. The research group examined all of them.
“One of the substances, ceritinib, also blocks the conversion of ATP by CD39,” Schäkel noted.
“We were able to show this not only in the test tube, but also in cultures with so-called triple-negative breast cancer cells. These are extremely difficult to treat – they usually hardly respond to therapies.”
Nevertheless, Müller does not think it makes sense to simply administer ceritinib as a CD39 inhibitor in certain cancers.
“After all, the active ingredient is primarily directed against a different group of enzymes; it would therefore have undesirable side effects,” she said.
“We now want to modify it so that it hardly inhibits protein kinases at all and instead slows down CD39 even more.”
Use in patients
Such an optimized active ingredient could also be combined with other therapeutic agents. “Classic cytostatics usually massively weaken the immune system; CD39 blockers, on the other hand, would activate it,” said Müller, who is also a member of the Transdisciplinary Research Areas (TRA) Building Blocks of Matter and Life and Health.
“In combination, therefore, the drugs could possibly have a significantly greater effect.”
Before use, it would first be possible to measure whether the cancer cells of affected patients actually carry a lot of CD39 on their surface.
“Because only then would treatment with CD39 inhibitors make sense,” Müller explained.
“So you would tailor the administration to the individual patient. This personalization of therapies for the purpose of enhancing efficiency is becoming increasingly important in medicine.”
Oncology R&D trends and breakthrough innovations