Newsletter Signup - Under Article / In Page
"*" indicates required fields
By Victoria Zazulina, M.D., head of development unit, oncology, for the healthcare business of Merck
Since 2000, February 4 has been recognized as World Cancer Day and marked by efforts across the globe to raise awareness of the disease’s devastating impact and encourage efforts to improve prevention, detection and treatment.
Over the past two decades, advances in diagnosis and treatment have contributed to declining cancer deaths and made survivorship possible for millions of people worldwide. But as long as people are still suffering and dying from cancer, we must strive to accelerate the discovery, development and delivery of innovative oncology medicines.
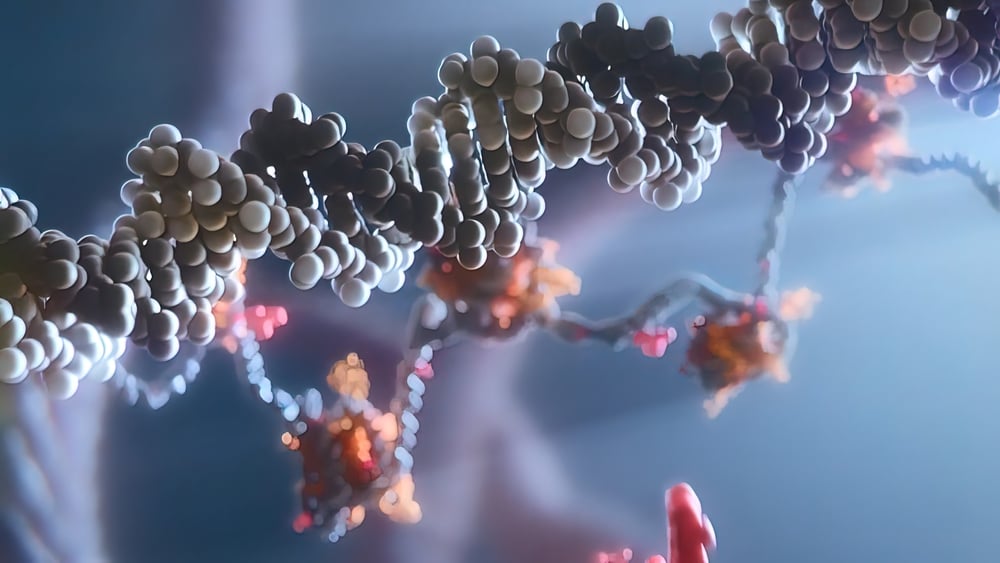
Collectively, we have made significant progress in understanding the biology of cancer, which is helping us uncover and exploit increasingly precise ways to defeat cancer cells. Many of the features that give rise and advantage to cancer cells can be directly linked with cells’ abnormal genetic makeup—their DNA. This is why mutations and genomic instability are key hallmarks of cancer.
In a normal cell in the body, disruption of its DNA should trigger DNA repair, a complex process that detects DNA damage and allows time to fix the breakdown to maintain integrity of the genetic code.
However, cells may have damaged DNA repair mechanisms, and over time, they can accumulate abnormal DNA and become cancerous. Such cells on the one hand may have an advantage over the normal cells because they can divide with less control over the structure of their DNA. However, this advantage can soon become their weakness: when too much faulty DNA is unrepaired, it may become incompatible with life and lead to a catastrophe for the cell. This is what we are trying to achieve with the new class of molecules called DNA damage response (DDR) inhibitors, which are being developed to speed up the “catastrophic” phase for cancer cells, by switching off various key enzymes in DNA repair mechanisms.
Our first glimpse into the power of DDR inhibition came with the introduction of PARP (poly ADP-ribose polymerase) inhibitors. These medicines have offered significant advances in the treatment of certain types of breast, ovarian, and prostate cancers. We and our peers in the industry are now working to build on this success from multiple angles as we seek to amplify the benefits for a broader range of patients.
Beyond PARP: The next wave of DDR inhibition
While PARP inhibitors were the first DDR inhibitors to gain approval, PARP is far from the only kinase involved in the complex mechanisms of DNA damage response. Additional enzymes involved in DNA repair mechanisms include, for example, ATR (ataxia telangiectasia and Rad3 related), ATM (ataxia telangiectasia mutated) and DNA-PK (DNA-dependent protein kinase), and their inhibition alone or in combination with standard-of-care treatments or PARP inhibitors are being explored in clinical trials.
With each of these approaches, particularly in some tumors with pre-existing faulty repair mechanisms, we have the potential to deliver a double punch on cancer DNA repair that tips the balance towards cell death.
The power of combinability
To date, PARP inhibitors have been used primarily as monotherapy. Investigational DDR inhibitors being researched today show the potential for enhanced efficacy when combined with similar agents or with other standard therapies used for cancer treatment:
- Cytotoxic chemotherapy, the role of which is to disrupt cancer cell DNA, creates synergies with DDR inhibitors that do not allow cancer to repair its damaged DNA.
- Immunotherapies, which research has indicated may enhance the natural immune response to cancer cells with faulty and unrepaired DNA and increase their recognition by the immune cells.
- Other DDR inhibitors, which collectively can block several DNA repair pathways in parallel, leaving cancer cells less chance to escape.
Identifying the right regimen for each patient
The ambition for anyone working in oncology research is to identify patients most likely to respond to a given treatment regimen based on their unique tumor biology. This is called a precision medicine approach, and it fully applies to the field of DDR inhibitors. We already use information about some mutations that can predict cancers to be sensitive to DDR inhibitors, yet there is much more research going on in this field to deepen our understanding of cancer vulnerabilities, escape mechanisms and synergies between various DNA damage pathways to improve the therapeutic impact of this new class of drugs.
Of the many cancer treatment modalities being investigated today, inhibition of DNA damage response is one of the most exciting, taking us directly to the source of cancer—its faulty and unrepaired DNA.
To learn more about World Cancer Day and how Merck is uniting our voice with the cancer community, see: Cancer Doesn’t Care, But We Do.
Oncology R&D trends and breakthrough innovations