This article was originally published in May 2016 authored by Dani Bancroft. It has since been updated to reflect the latest developments in the autoimmune disease field.
While we mostly think of disease as being caused by bacteria or viruses, an illness can often occur when the body’s own defenses turn against itself. Here we’ll take a look at seven rare autoimmune diseases you may not have heard of and what biotechs are doing to treat them.
The body’s immune system can be equally as toxic to foreign organisms as it can to its own tissues. A hyperactive immune system can wreak havoc on the body, with patients often afflicted by more than one autoimmune disease at once.
We discuss many well-known rare autoimmune diseases on a regular basis, such as multiple sclerosis, which results from an immune attack on the myelin sheath of nerve cells; type 1 diabetes, where the immune system destroys insulin-producing pancreatic cells; rheumatoid arthritis, in which the joints suffer from inflammation; and lupus, when the whole body is under attack from the immune system.
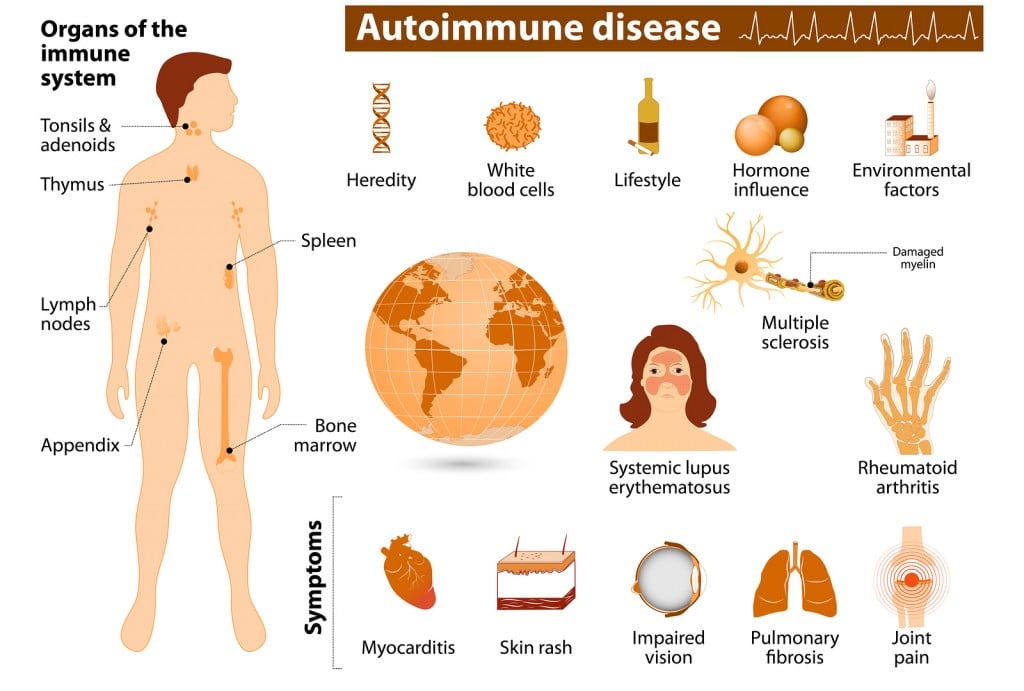
There are many other rare autoimmune diseases that also take a great toll on patients. We’ve reviewed seven of these rare conditions that biotech companies are also fighting to treat.
Table of contents
Grave’s Disease
There are different types of rare autoimmune diseases that cause inflammation of the thyroid gland, which can lead to hormonal imbalances. Grave’s disease occurs when antibodies induce the thyroid to secrete too much thyroid hormone, which can lead to tremors, red skin and an irregular heartbeat.
Affecting 2-3% of the world’s population, Grave’s disease is typically treated by removing the thyroid gland and giving patients lifelong hormone replacement therapy. However, synthetic thyroxine can induce hypothyroidism, a condition where the thyroid gland is not active enough. Due to this, new treatment methods have been pivotal to tackle the disease.
In 2022, the Food and Drug Administration (FDA) approved Horizon Therapeutics’ Tepezza for the treatment of active Graves’ orbitopathy in adults after it had shown significant results in clinical trials, particularly in its ability to reverse proptosis, a condition that affects the vision of Grave’s disease patients.
Earlier in 2018, British-Belgian biotech company Apitope obtained positive phase I results, for a treatment that uses a protein to inactivate white blood cells that attack the thyroid gland. When the trial was completed, most of the patients had a properly functioning thyroid gland with normal levels of thyroid-stimulating hormone receptor antibody.
Psoriasis
A common misconception of dermatological conditions like psoriasis is that they only cause cosmetic discomfort. However, similar to burn victims, the skin damage can be very serious during flare-ups of the disease. This results in the inability to regulate body temperature which makes psoriasis patients vulnerable to life-threatening infections. Today, palliative care and biologics, which block cells or proteins involved in the disease, are available to treat psoriasis. However, antibody treatment may prove to be a more effective approach, and biotechs are racing to bring their treatments to the market.
In May 2022, the FDA approved Tapinarof cream 1%, developed by clinical-stage company Dermavant Sciences for plaque psoriasis which causes dry skin lesions. The steroid-free topical treatment targets gene expression and reduction of inflammatory TH17 cytokines and TH2 cells, which are cell signaling proteins.
The first oral therapy, Deucravacitinib manufactured by pharma company Bristol Myers Squibb, was also approved by the FDA for the treatment of moderate-to-severe plaque psoriasis in adults. This followed successful clinical trials where a majority of the patients obtained a Psoriasis Area and Severity Index of 90, indicating more than 90% improvement from the baseline.
The Danish LEO Pharma also received approval from the European Medicines Agency (EMA) for its antibody Kyntheum (brodalumab). Following closely behind, for its antibody guselkumab, German biotech MorphoSys was approved by the FDA for the treatment of moderate to severe plaque psoriasis in 2017, and active psoriatic arthritis in 2020.
Other companies working in the psoriasis field include Delenex Therapeutics, Sweden-based Affibody, and Switch Biotech in Germany.
Uveitis
Uveitis is a rare autoimmune disease that causes inflammation of the pigmented layer of the eye, called the uvea, the optic nerve and the gel that gives the eye its shape, known as the vitreous. Uveitis, which can lead to eye infection and pain, is usually treated with anti-inflammatory topical, intravenous and oral corticosteroids.
As with psoriasis, many biotechs are hedging their bets on new treatments for uveitis, and some have already been successful. In October 2021, the FDA approved xipere, a therapy to treat macular edema associated with uveitis, which is characterized by the accumulation of fluid in the eye. Administered via injection, the drug demonstrated success in clinical studies. The therapy is a collaboration between healthcare companies Bausch + Lomb and Clearside Biomedical Inc.
This article was originally published in May 2016 authored by Dani Bancroft. It has since been updated to reflect the latest developments in the autoimmune disease field.
Moreover, as cell therapies gain traction, biotechs have used technology to treat uveitis. TxCell based in Nice, France, was granted EU and US Orphan Drug Designation for its T-cell based therapy for autoimmune uveitis in 2015. Last year, Galapagos and Gilead also launched filgotinib, which was successful in reducing the risk of flare in uveitis patients after a successful phase 2 clinical study.
Sarcoidosis
Sarcoidosis causes inflammatory cells to build up in certain tissues, particularly the lungs, to form granulomas. Symptoms of the autoimmune condition include shortness of breath and bumps on the skin, and sometimes when other organs are affected, can lead to irregular heart rhythm, also known as arrhythmia.
One of the latest treatments to receive fast track designation from the FDA is aTyr Pharma’s Efzofitimod, a drug that downregulates the immune response in inflammatory conditions. This came after positive phase 1b/2a trials in 2022, and has begun phase 3 studies in sarcoidosis patients.
Other therapies include Janssen’s antibody Remicade (infliximab), which has been effective in treating refractory sarcoidosis. AbbVie’s Humira can also be effective in treating patients who have not responded well to standard sarcoidosis treatments.
Addison’s
Addison’s disease affects the endocrine system, which controls the release of hormones. It is caused when the adrenal glands are damaged, resulting in its inability to produce sufficient amounts of the hormones cortisol and adrenaline. This can cause a range of symptoms including weakness, pain, low blood pressure (due to salt loss) and hyperpigmentation in areas of the skin. Acute attacks can also occur when the hormone levels drop too low.
Addison’s disease is treatable with regular hormone replacement therapy to replace cortisol. However, this can be cumbersome since, like diabetics, patients have to carry around syringes of cortisol for emergency use.
In 2018, researchers at Queen Mary University of London used cells in urine to begin generating an artificial adrenal gland, which could help treat Addison’s disease by replenishing the body’s cortisol levels.
Vitiligo
Vitiligo is often mistaken for hyperpigmentation of the skin, but is in fact the opposite — the loss of skin pigment. It is commonly treated with corticosteroids and light therapy, which can lead to premature skin aging and increase the risk of skin cancer. Long-term corticosteroid use has a range of side effects, from osteoporosis to increased risk of infection and diabetes. Therefore, finding alternative treatment options can only benefit patients, and European biotechs have some promising candidates.
In 2022, the FDA approved the first at-home treatment for this rare autoimmune disease. Global biopharma Incyte’s ruxolitinib cream Opzelura, is a topical therapy that targets the overactive immune system and helps in the growth of healthy skin cells to restore pigment to the area. The treatment performed well in clinical trials where 30% of the participants had regained at least 75% skin repigmentation on their faces in 24 weeks, which increased to 50% of people after six months.
Other treatments for vitiligo are on the horizon as well. Currently, studies on nanoparticle drug-delivery technology are underway. Meanwhile, the drug afamelanotide for repigmentation therapy, is being evaluated by Australia-based biotech Clinuvel Pharmaceuticals following approval by U.S and European regulators, as of 2022.
Granulomatosis
Granulomatosis is a type of vasculitis, or inflammation of blood vessels, which encompasses many syndromes and rare autoimmune diseases, some of which are poorly understood. Granulomatosis can lead to weight loss, fatigue and shortness of breath. It is commonly treated with corticosteroids and immunosuppressant drugs, which can increase the risk of infection.
One treatment option for granulomatosis patients is rituximab, a therapy that is administered through injection, and is often combined with corticosteroids.
In 2021, the FDA approved Tavneos, an add-on therapy acquired by U.S-based biopharma Amgen, that targets pro-inflammatory proteins to decrease blood vessel damage. The clinical trials were met with positive efficacy results, however, some adverse side-effects were noticed, which included nausea and vomiting.
Previously, University of Groningen in the Netherlands along with U.S biotech Kineta were developing a drug derived from sea anemones that was found to have had a positive effect on the complications derived from granulomatosis, among other rare autoimmune diseases.
As more people are diagnosed with rare autoimmune diseases worldwide, developing new treatments could help decrease the toll these conditions take on the quality of life and reduce side effects from other therapies. While many biotechs are focusing their efforts on antibody treatments, other technologies like cell therapy are also paving the way forward in fighting rare autoimmune diseases.