Newsletter Signup - Under Article / In Page
"*" indicates required fields
Chronic metabolic disorders like type 2 diabetes and cardiovascular disease are a huge burden on healthcare systems, and their treatment is often fragmented and reliant on trial-and-error approaches. Rebecca Cripps and Robert Thong at MultiOmic Health explain how precision medicine is set to change the way we think about these disorders.
Metabolic disorders constitute the world’s largest health burden, with one in three adults showing signs of metabolic syndrome – a combination of risk factors including faulty blood sugar regulation, excess weight, hypertension and high blood levels of lipids such as cholesterol and triglycerides. These risk factors are the root cause of chronic age-related conditions such as atherosclerotic cardiovascular disease, type 2 diabetes (T2D), chronic kidney disease (CKD), non-alcoholic fatty liver (NAFLD/NASH) and polycystic ovary syndrome.
Together, contributing nearly half the world’s fatalities, chronic metabolic conditions ran up a global healthcare tab of almost $2 trillion in 2019. With tailwinds such as population aging, fast food diets and sedentary lifestyles, their global cost burden has been forecast to exceed $5 trillion in 2040.
The COVID-19 pandemic will simply worsen this estimate. A substantial proportion of Covid hospitalizations and deaths happen in patients with metabolic syndrome and emerging evidence suggests Covid survivors are at an increased risk of eventually developing both T2D and CKD.
Current treatments are neither precise nor cures
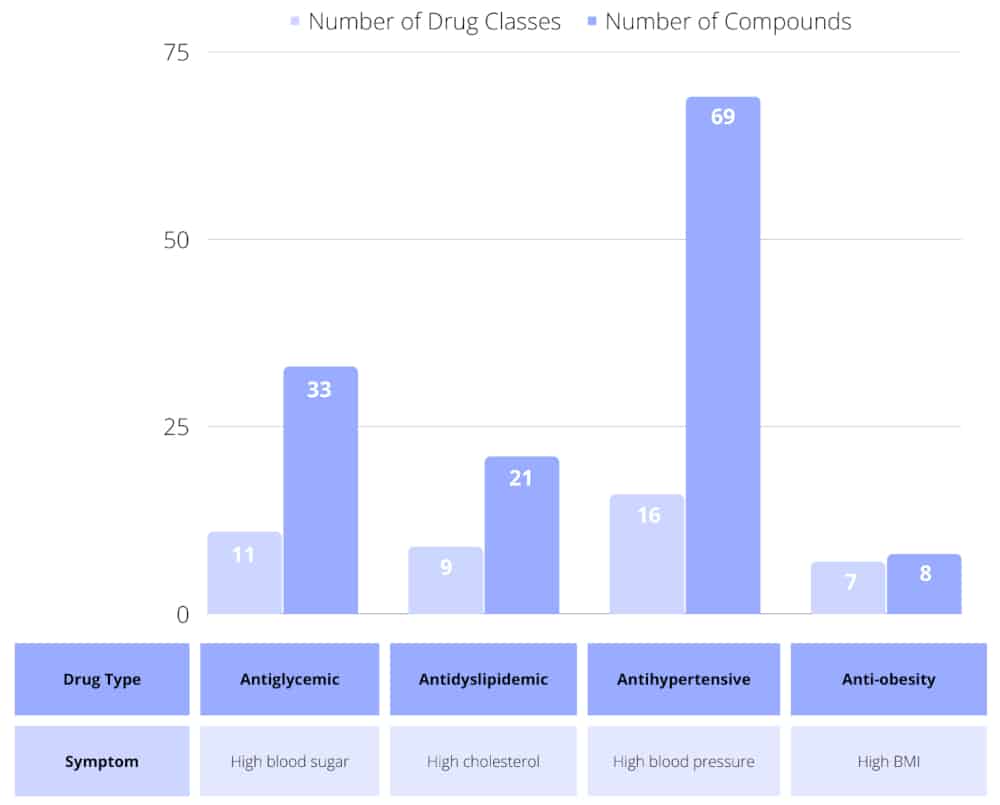
Therapeutic strategies for these diseases are currently one-size-fits-all, treating individual symptoms with drug classes selected according to age and ethnicity. There are many distinct drug classes, each with multiple drug options approved to treat individual symptoms. The right cocktail of specific drugs that work best for someone has to be determined via trial and error. If symptoms remain uncontrolled, selected drug classes are escalated from first- to second- and then to third-line options.
Consider hypertension: there are 16 drug classes with a total of 69 individual approved drugs. The first-line treatment strategy utilizes one drug from either the angiotensin-converting enzyme inhibitor, angiotensin receptor blocker or calcium channel blocker classes. If this fails to control the patient’s blood pressure, a combination of drugs from two of the three classes is tried. And if that fails, the third-line strategy adds a thiazide-like diuretic to the mix. Furthermore, if the patient also has high blood lipids, then the whole scenario is replayed in parallel with different dyslipidemia drug classes.
Current therapeutics merely reduce the risk of – rather than eliminate – serious downstream complications such as heart attack, stroke, blindness, nerve damage, foot amputation, kidney dialysis and liver failure. A significant proportion of patients treated according to medical guidelines eventually develop life-changing complications.
For example, statins are prescribed to patients with high levels of low-density lipoprotein cholesterol to avoid cardiovascular incidents. A recent scientific study reported that 44% of cardiovascular patients taking statins nevertheless experience their first major adverse cardiovascular event within seven years.
Additionally, many patients present with overlapping disease traits and multiple diseases but different medical specialists – cardiologists, endocrinologists, nephrologists, hepatologists – treat individual symptoms. Treatment strategies are therefore compartmentalized, with some patients having to self-manage information flow between specialists.
The need for precision interventions
A new disease taxonomy is emerging in leading academic medical circles, propounding that each patient has one disease that takes them on a journey of various symptomatic manifestations (e.g., T2D, CKD) and downstream complications (e.g., heart attack, kidney damage).
Although huge diversity is evident in patient journeys, many consistent patterns are observed. Each pattern represents a clinical “phenotype,” namely a patient sub-group experiencing a similar journey. As documented in cancer, patients within a phenotype may be subdivided into different patient “endotypes,” each endotype reflecting a different set of underlying disease drivers such as certain genetic mutations.
The essence of precision medicine is selecting the optimum treatment for each endotype. For cancer, there are over 80 precision medicines for nearly 40 currently recognized endotypes. To slow or reverse disease progression in metabolic disorders, different therapeutic strategies are also needed for each patient endotype.
Precision medicine prevents the waste of healthcare resources on treating patients with sub-optimal medications, which can increase their likelihood of serious complications. Well-characterized endotypes, each with their own optimal treatment strategy straddling traditional symptomatic classifications, will also ensure an integrated care approach combining the different medical specialists.
Searching for metabolic endotypes at the molecular level
While a clinical phenotype is definable using observed clinical traits and standard diagnostic tests, uncovering the complex interplay of multiple factors underlying a metabolic syndrome endotype requires operating at the molecular level.
Other than a few rare genetic diseases, metabolic syndrome endotypes cannot be understood by looking purely at genetic mutations in the part of our genome that codes our proteins. Which proteins actually get made in the cell, the modifications these proteins undergo and the influence of our metabolic processes all contribute to an endotype’s character.
To define and understand the endotype behind an observed clinical phenotype, we need to examine multiple molecular “omics” factors, including the non-coding DNA, epigenome, transcriptome, proteome and metabolome. It is only in recent years that researchers have been able to affordably generate this kind of data, and the computing power and predictive analytics tools needed to interrogate the huge resulting datasets.
R&D and partnering activity heating up
Now that detailed omics is accessible to researchers, R&D activity is exploding. Across the metabolic syndrome conditions combined, the number of scientific papers published increased by 70% from 2010 to 2021, compared to a 52% increase for cancer.
Momentum is also building in biopharma R&D; 11 of the top 17 pharmas have cardiovascular, renal or metabolic disease as one of their core research areas. Recently, Merck/MSD revealed plans for cardiovascular drugs to once again be a large component of its future R&D activity. Novo Nordisk also set itself a goal to expand its pipeline into disease areas adjacent to diabetes, such as cardiovascular and metabolic disease, obesity and NAFLD. As a company with a long-standing sole focus on diabetes, this is a clear change in strategy.
Addressing disease drivers and patient endotypes through the application of innovative emerging technologies such as multiple omics modalities, artificial intelligence and high throughput human cell- and tissue-based experiments will kick-start precision medicine development for metabolic disorders. A key driver of this progress will be collaborations between established pharmas and startup biotechs to rejuvenate traditional R&D and bring to market transformational medicines in the metabolic disorders arena.

Robert Thong is CEO of MultiOmic Health. He has over 30 years’ experience across the bioscience sector specializing in research-based business models, business development and R&D management.

Becky Cripps is Business & Science Analyst at MultiOmic Health. She has spent the last two years analyzing the metabolic syndrome-related therapeutics space.






