Newsletter Signup - Under Article / In Page
"*" indicates required fields
Agendia will launch a next-generation sequencing test in the EU to determine the chances of metastasis in breast cancer patients.
Agendia, a US-Dutch biotech specializing in personalized medicine and molecular diagnostics, has obtained CE marking for the MammaPrint BluePrint Kit, a diagnostic device for breast cancer that uses next-generation sequencing technology. The kit can help clinicians identify women with breast cancer who are at high risk of having their cancer spreading to other tissues.
The new kit combines two different Agendia tests already in the market. The first, called MammaPrint, analyzes 70 genes to determine the risk of cancer recurrence. The second, called BluePrint, analyzes 80 genes to classify the molecular subtype of cancer. This information can then be used to select a personalized treatment for each patient.
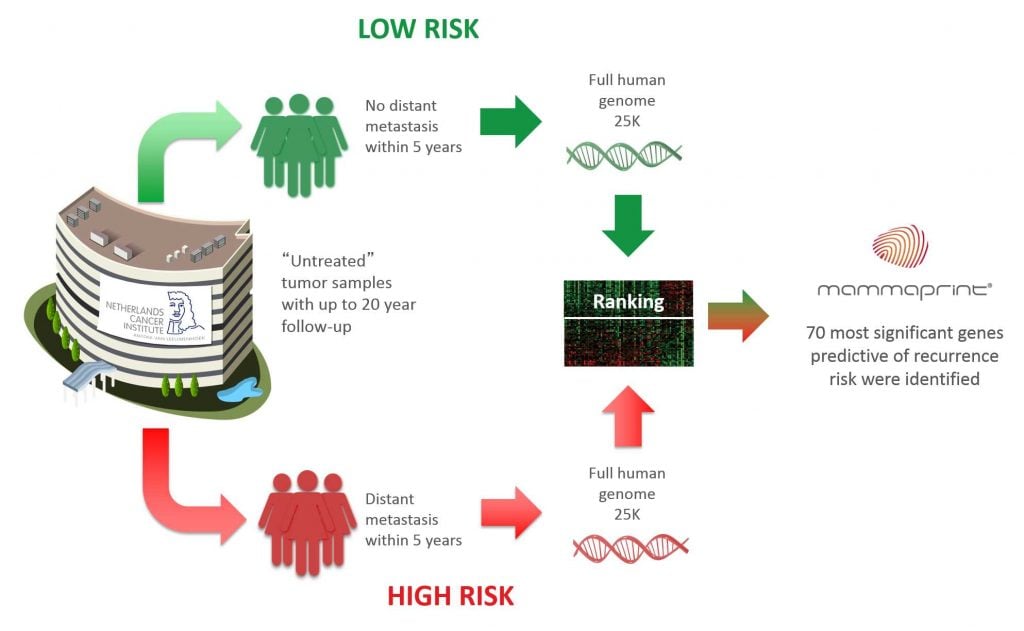
Unlike Agendia’s previous tests, the new kit sequences the patient’s RNA instead of the DNA. This approach offers a more accurate diagnosis by measuring the expression of the genes rather than only their sequence.
Agendia’s MammaPrint test has proven that it can prevent the unnecessary use of chemotherapy in 46% of breast cancer patients. The new test builds on this concept by adding an analysis of the molecular profile of the patient’s cancer that can help determine what’s the best alternative to chemo for each patient.
Seeing that there are nearly 500,000 new cases of breast cancer reported every year, targeting treatments such as chemotherapy effectively could reduce costs and improve patient quality of life.
Images by Agendia, Photographee.eu/Shutterstock
Oncology R&D trends and breakthrough innovations







