Newsletter Signup - Under Article / In Page
"*" indicates required fields
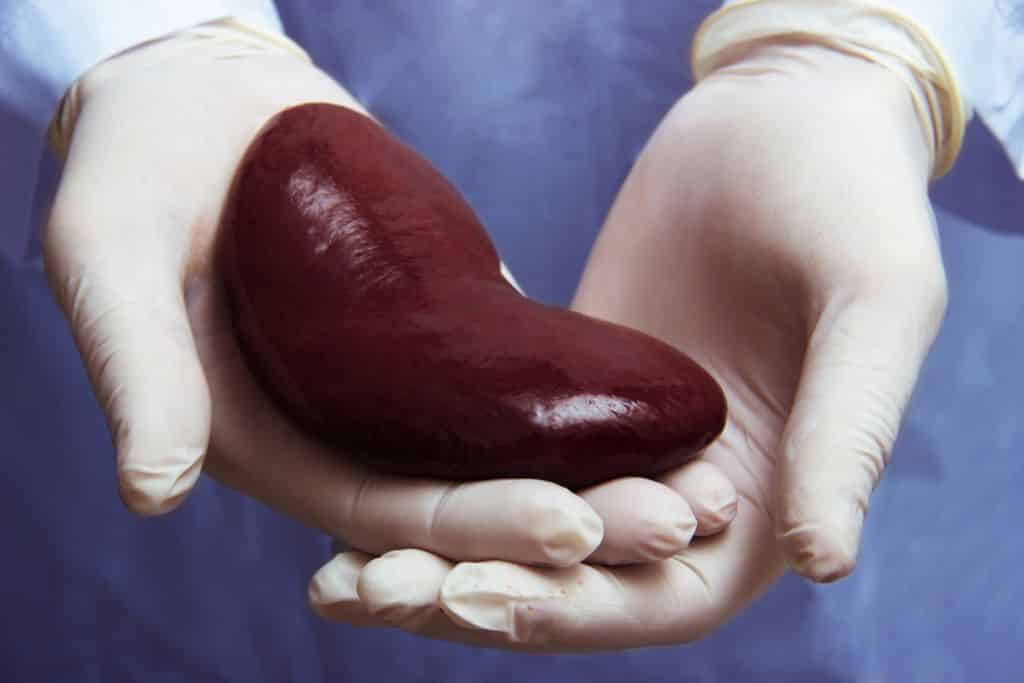
Treatment offered by Hansa Biopharma for the densensitization of highly sensitized adults prior to kidney transplants from deceased donors has been recommended by the UK’s National Institute for Health and Care Excellence (NICE).
NICE considers Idefirix (imlifidase), made by Hansa, a Swedish company that specializes in enzyme technology for rare immunological conditions, to be a clinically and cost-effective treatment.
Candidates for a kidney transplant are considered highly sensitized if they have pre-formed antibodies against available donors known as human leukocyte antigens (HLA).
These antibodies can carry the risk of causing damage to the transplanted kidney and potentially lead them to rejection. Risk factors for becoming highly-sensitized include previous transplants, blood transfusions and pregnancy. Finding a match for these patients can prove difficult and can mean they spend longer on waiting lists, which increases their risk of dying before a match is found.
Kidney transplants
The annual average number of kidney transplants in the UK over the last five years is 2,400 from deceased donors and 926 from living donors.
Imlifidase is an enzyme derived from the bacterium Streptococcus pyogenes and has the ability to specifically target and cleave all classes of immunoglobin G (IgG) antibodies. It is a promising new strategy for desensitization of transplant patients with donor-specific anti-HLA (human leukocyte antigens) antibodies.
Highly sensitized patients have high levels of these preformed antibodies that can bind to the donor organ and damage the transplant. However, once they are activated with imlifidase, there is a window of opportunity for the transplant to take place. By the time the body starts to synthesize new IgG, the patient will be receiving immunosuppressive therapy to reduce the risk of organ rejection.
Adnan Sharif, trustee at Kidney Research UK, said: “A lack of effective desensitization approaches has meant that until now, people who are classed as highly sensitized kidney patients in England have struggled to find a match and have often had no alternative but to remain on long-term dialysis with a very poor quality of life.
“We welcome this decision, which will allow new opportunities for certain highly sensitized patients to qualify for a life-altering transplant and gain freedom from dialysis.”
Pre-transplant treatment
The efficacy and safety of imlifidase as a pre-transplant treatment to reduce donor-specific IgG was studied in four phase 2 open-label, single-arm, six-month clinical trials.
Hansa is now collecting further clinical evidence and will submit additional efficacy and safety data based on one observational follow-up study and one post-approval efficacy study.
Idefirix was reviewed as part of the European Medicines Agency’s (EMA) PRIiority MEdicines (PRIME) program, which supports medicines that may offer major therapeutic advantage over existing treatments or benefit patients without treatment options.
The first application for EMA market approval for the kidney transplant drug, was made by Hansa Biopharma in 2019.
Cover image: Shutterstock
Artificial intelligence and the future of antibody discovery