Genmab’s leukemia drug will be available around the world through compassionate use programs, while it will continue to be marketed as normal in the US.
Blockbuster drug owner Genmab developed Arzerra for the treatment of chronic lymphocytic leukemia (CLL) but the drug will be taken off the market outside the US due to its low uptake. Novartis markets the drug and will transition it from commercially available to limited availability through compassionate use programs outside the US, meaning in-need patients will continue to have access to it.
Novartis will pay Genmab $50M (€41M) to cover any lost milestones and royalties, which could be put towards other exciting candidates in its pipeline like daratumumab, which achieved promising Phase III results last year. However, this has not stopped the company’s stock price falling by 5% since the news.
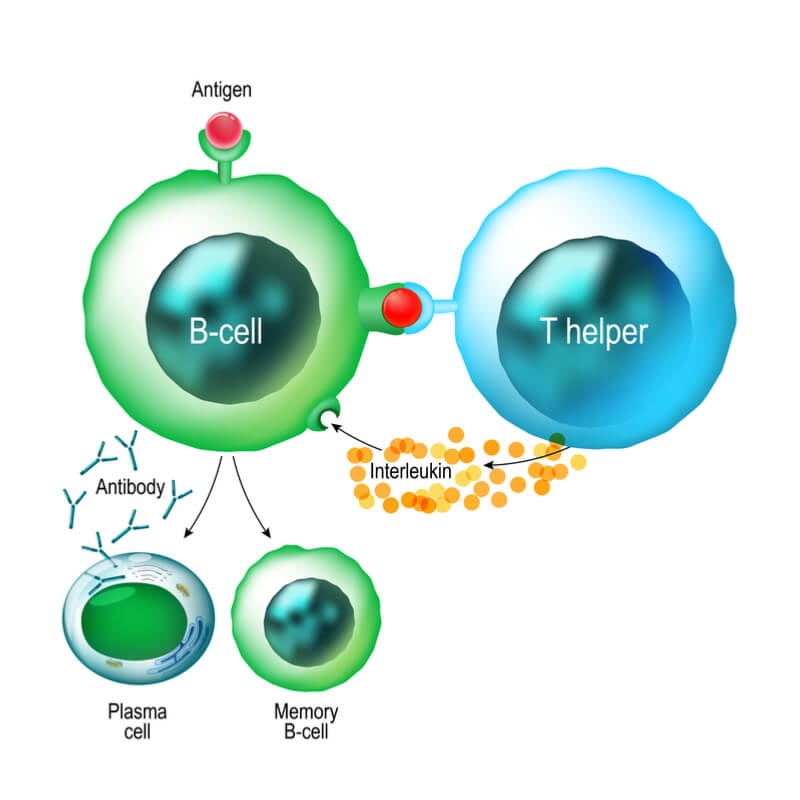
Each year, there are almost 21,000 CLL cases and the disease is responsible for around 4,500 deaths in the US alone. Various forms of leukemia affect different blood cells, and in the case of CLL, the disease affects B cells. Arzerra, or ofatumumab, is a monoclonal antibody that targets CD20 proteins on the surface of leukemia cells. The antibody binds to the proteins, alerting the immune system, which can then hunt down and kill the cancer cells.
Genmab CEO Jan van de Winkel explained the big pharma company’s decision to transition the drug: “[The decision] reflects the fact that many more drugs have become available for CLL over the last five years and that there is a low number of patients using Arzerra outside of the US market. The compassionate use programs will ensure that patients who benefit from Arzerra can remain on treatment.” However, in the US, uptake remains high so Novartis is willing to continue providing the drug in this market.
Novartis will begin the transition process as soon as possible, once a compassionate use program has been structured with regulatory bodies that ensures that patients benefiting from Arzerra receive the drug for as long as they need it, free of charge. However, this is not the end of the road for the drug, with clinical studies investigating its use for multiple sclerosis and indolent non-Hodgkin lymphoma set to continue.
Current treatment options for the disease include chemotherapy, targeted therapy, and stem cell transplants, as well as monoclonal antibodies. Two alternative monoclonal antibodies, rituximab and obinutuzumab, are often preferred to Arzerra, which is often used as a final attempt to treat unresponsive cancers. This is also without considering new therapies that will be entering the leukemia field in the near future. Last year, we saw the approval of two CAR-T cell therapies, Novartis’ Kymriah and Gilead’s Yescarta. With both companies intending to take their technology to new indications, CLL may well be a disease that they investigate next.
With other drugs available to CLL patients, it seems a sensible decision for Novartis to pull Arzerra out of the market. Although the decision may be driven by money, it is encouraging to see the company keeping the drug available to patients that benefit from its use. Perhaps the money that is saved can be used to develop the drug for other diseases or new therapies for CLL.
Images – Medtech THAI STUDIO LAB 249 / shutterstock.com; Designua / shutterstock.com