Newsletter Signup - Under Article / In Page
"*" indicates required fields
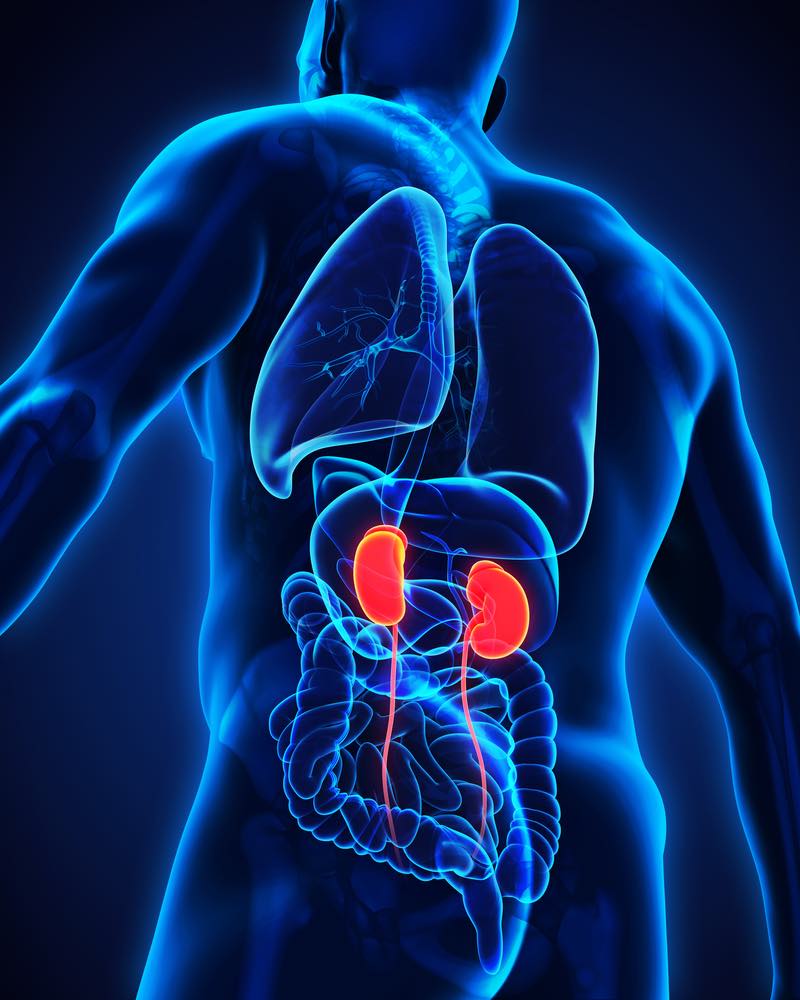
The first and only approved treatment for primary immunoglobin A (IgA) nephropathy, a rare, progressive disease of the kidneys, has been launched in Germany.
European commercial partner of Calliditas Therapeutics AB, STADA Arzneimittel AG, says the treatment will launch in additional European countries later.
The disease, also known as Berger’s disease, occurs when immunoglobin builds up in the kidneys which results in local inflammation which over time can hamper the kidneys’ ability to filter waste from the blood.
“We are excited that STADA is in the position to swiftly launch this product in Europe, starting with the German market. To bring an approved medication to patients suffering from this rare disease has been our focus since we started this endeavor well over a decade ago,” said Calliditas CEO Renée Aguiar-Lucander.
Only approved treatment
Calliditas received conditional approval in July from the European Commission for the development candidate Nefecon, providing the first and only approved treatment alternative for adult patients with IgAN at risk of rapid disease progression with a urine protein-to-creatinine ratio (UPCR) ≥1.5 g/gram.
The conditional marketing authorization, which has now been transferred to STADA Arzneimittel, applies in all 27 European Union Member States as well as Iceland, Norway, and Liechtenstein. This is the first orphan medicine introduced through STADA Arzneimittel’ Specialty Care business unit.
STADA Arzneimittel CEO Peter Goldschmidt said: “Making this product available to primary IgAN patients in Europe brings for the first time a therapeutic option to an under-served patient population.
“The launch of STADA’s first orphan Specialty medicine is evidence of how STADA is bringing additional value to patients, healthcare professionals and health systems through a broad portfolio of Specialty, Generics and Consumer Healthcare products.“
Germany: STADA Arzneimittel’s lead launch market
In Germany, the lead launch market, 3.1 people per 100,000 develop IgAN each year, a frequency slightly higher than the 2.5 per 100,000 which is the estimated global incidence.
Calliditas Therapeutics is a commercial stage biopharma company based in Stockholm, Sweden focused on identifying, developing and commercializing treatments in orphan indications, with an initial focus on renal and hepatic diseases with significant unmet medical needs.
Calliditas’ lead product, developed under the name Nefecon, has been granted accelerated approval by the FDA under the trade name TARPEYOTM and conditional marketing authorization by the European Commission under the trade name KINPEYGO.
STADA Arzneimittel is headquartered in Bad Vilbel, Germany. The company focuses on a three-pillar strategy consisting of generics, specialty pharma and non-prescription consumer healthcare products.