Newsletter Signup - Under Article / In Page
"*" indicates required fields
Verona Pharma obtained positive Phase IIa trial results for a unique cystic fibrosis drug that inhibits two disease-related enzymes simultaneously, showing promise for treating a high unmet patient need.
Verona Pharma, based in London, UK, aims to improve the lives of people with chronic respiratory diseases. Verona states their Phase IIa data provide grounds to further develop their drug, RPL554, for the treatment of cystic fibrosis. The drug uses a unique mechanism of action that allows it to target the disease in multiple ways.
In patients with cystic fibrosis, a defective gene causes mucus secretions in the lungs to become sticky and thick. As a result, patients respiratory systems become plugged up. Current treatments for cystic fibrosis include antibiotics, drugs that clear mucus, bronchodilators to make breathing easier, as well as corticosteroids to reduce inflammation.
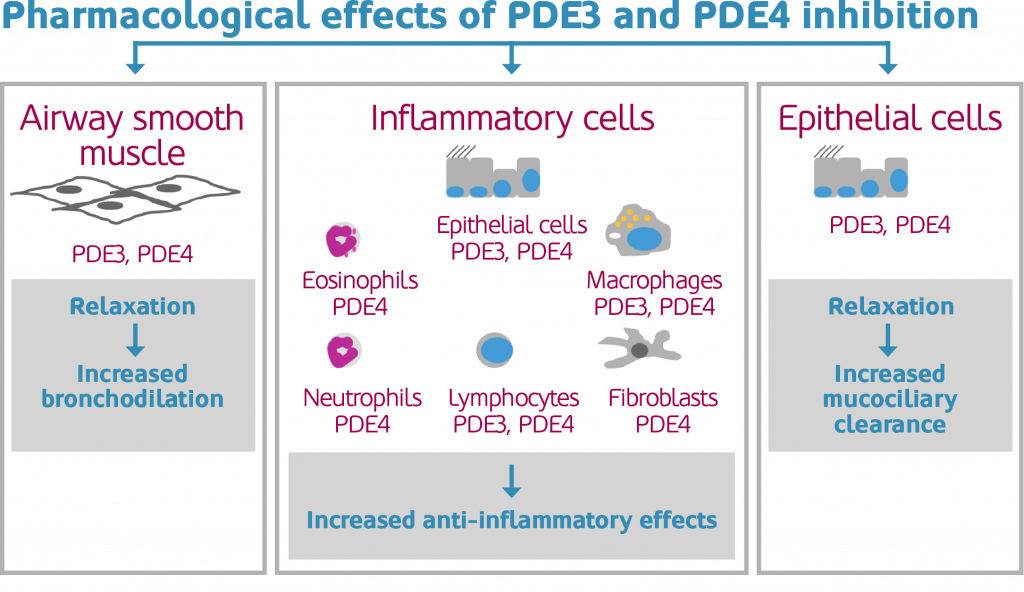
RPL554 is an inhaled treatment that acts as both a bronchodilator and an anti-inflammatory agent, a feat no other available treatment has been able to match. The drug achieves these two functions through a unique mechanism of action. By using a dual inhibitor, the drug can target two disease-related enzymes (PDE3 and PDE4) simultaneously.
Other treatments for cystic fibrosis are in development as well. Galapagos and AbbVie are collaborating on small molecule drugs for cystic fibrosis that could restore the function of the CTFR chloride channel, which is defective in cystic fibrosis. French biotech Antabio was awarded $8.9M (€7.6M) by CARB-X to develop small molecule antibiotics against Pseudomonas aeruginosa infections, a common risk to cystic fibrosis patients. Unlike Verona’s, these treatments have a single function or target.
Additionally, research could bring biotech’s closer to developing cystic fibrosis treatments that move beyond symptom control to correcting disease causes. For example, researchers from the University of Zurich have found a new chloride channel that could be used as an alternative drug target to CTFR in developing new treatments.
Cystic fibrosis is a rare hereditary disease of the lungs affecting approximately 70,000 people worldwide. Despite its rare occurrence, the global cystic fibrosis therapeutics market is estimated to reach $13.9Bn (€11.32Bn) by 2025. Time will tell if Verona’s unique pharmacological approach will give it a competitive market position.
Images by Verona Pharma, Magic mine/Shutterstock