Newsletter Signup - Under Article / In Page
"*" indicates required fields
“Plasmids are critical for so many processes in early drug discovery,” says David Öling, associate principal scientist at AstraZeneca.
“From protein production, cell line engineering, CAR-T cell therapy, viral vector generation, therapeutic genome editing, mRNA/vaccine production and more. The time it takes and the cost of producing plasmids for DNA constructs is a critical limiting factor.”
This challenge became particularly poignant during the COVID pandemic, when disruptions to supply chains and staffing limited access and delayed timelines even more.
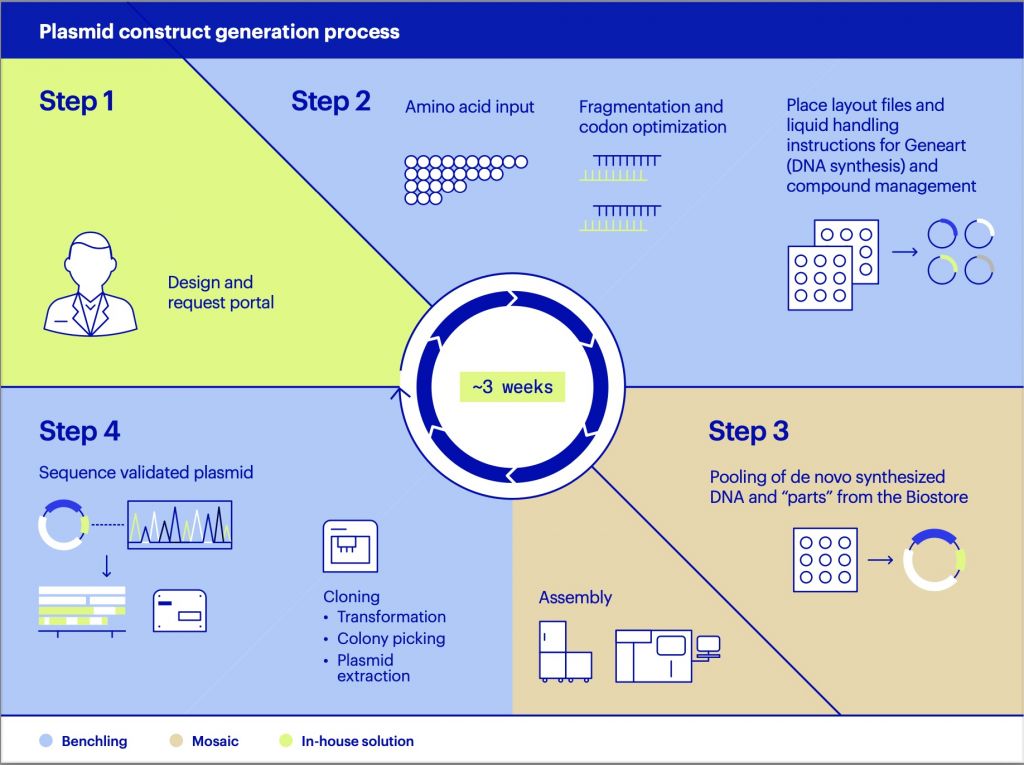
But that’s all changing. A combined team of computational and molecular biologists at AstraZeneca and tech operators at Benchling succeeded in creating a novel workaround for the whole DNA synthesis industry, and one that they’ve made open-sourced, available now on GitHub. It’s called FRAGLER — FRAGment recycLER. Their solution takes three weeks for plasmid construction at scale and led to cost savings from 50 to 90%.
“This is a timeline no DNA synthesis vendor can equal,” said Öling, “particularly for long, complex sequences such as SarsCoV or Cas9 expression constructs.”
Tell us about FRAGLER and how you thought to introduce sequence recycling into the process?
Traditionally, a significant amount of synthesis time and cost is spent on DNA sequences that are present multiple times across a construct, as those sequences have to be ordered individually as many times as they are present. But what if those sequences could be recycled, reused?
To pull this off, we would need to create an algorithm for fragment recycling which identifies the shared sequences across constructs so that they need to be ordered once, reducing both the cost of DNA synthesis and the time it takes to generate DNA fragments.
With our FRAGLER solution, we essentially created software that increases efficiency and throughput of DNA synthesis on a wide variety of targets. FRAGLER not only performs amino acid sequence alignment and codon optimization of desired sequences, but it also fragments those sequences to increase the success of DNA production. Fragmentation of long sequences into shorter fragments of 300-900 base pairs reduces synthesis time and ensures production success. The other benefit is that fragmentation eliminates sequence complexities and minimizes cancellations, which are very common for longer sequences.
What were the origins of this idea? How long did it take to come to fruition?
My team provides most of the DNA constructs used by AstraZeneca, Discovery Biology across Sweden and the U.K. The long timelines we experienced with having to outsource DNA fragments was something we knew could be optimized and modernized. Then Covid acted as a catalyst — where timelines and urgency were highly pressurized.
With FRAGLER, we were going from fully outsourcing to fully insourcing, so there were a lot of learnings along the way. Conceptually, the idea worked quickly. It was really about optimizing so we could ensure quality while driving down timelines and costs – the role of bioinformatics was really critical here.
The build process began in 2020, and that’s when we started to work with Benchling to create an automated DNA search, fragmentation and assembly workflow.
What were some of the challenges you ran up against with productizing FRAGLER?
While you can run FRAGLER offline, we found that this didn’t provide the scale we needed to utilize the full potential of the tool. Thousands of constructs are generated during early stage drug discovery, and no one human can reasonably keep track of this. It became apparent early on that for FRAGLER to be used effectively, we needed the ability to efficiently identify fragments for reusing, which is where Benchling came in.
Previously, we had been relying on manual processes with tools like Excel and various in-house applications to keep track of constructs. Not only was recording this time intensive, but limited how we worked together and shared findings. Implementing Benchling allowed us to automate most steps in this process, whilst also providing a single database for which thousands of pre-existing fragments can be rapidly searched through.
One of the keys to FRAGLER seems to be its ability to automate workflows – that it gets better at identifying duplicative DNA sequences over time, as more data is added to the system.
The first time you run a campaign, FRAGLER will identify duplicate DNA sequences and reduce the cost of synthetic DNA synthesis by up to 50% with fragment recycling. But for each campaign after that there is an estimated 10-30% additional cost reduction per iterative cycle, as more and more fragments are recycled and reused with each campaign.
Eventually, most DNA fragments will be available in-house, meaning that you’ll reach nearly zero cost for generation of new synthetic DNA constructs. This process relies heavily on the algorithmic sequence search as manual searches for duplicate sequences at this scale are impossible.
How will the advancement with FRAGLER benefit R&D? Who stands to benefit?
An essential first step in drug discovery is recombinant protein production. Expression and purification of the intended target protein enables candidate drug screening as well as structural and mechanistic interrogations that guide the design and evaluation of candidate drugs. Later, cell lines are engineered and used in in vitro screening to evaluate toxicity and confirm a candidate drug’s mode of action. These early tests are critical for eliminating ineffective and toxic drugs early, before billions of dollars are spent on clinical trials doomed to fail.
DNA constructs — artificially designed vectors or plasmids that deliver target DNA sequences into cells, where the gene product is produced— underpin recombinant protein production and cell line engineering. Rapid and flexible generation of these constructs at scale is one of the most limiting first steps in drug discovery. A large time sink associated with this process is the de novo synthesis of long coding sequences, a challenge that modular cloning kits haven’t been able to solve effectively.
To address this, our DNA assembly framework with FRAGLER, integrated into Benchling, supports rapid, cost- efficient, and scalable construct generation. We’ve reduced the window down to a robust 3 week process for codon optimized and template independent plasmid constructs.
Astrazeneca and Benchling have published a whitepaper on the automated assembly framework.
Partnering 2030: Biopharma Report







