Newsletter Signup - Under Article / In Page
"*" indicates required fields
Innate Pharma and MedImmune will test the combination of Innate’s cancer antibody and the checkpoint inhibitor durvalumab in patients with solid tumors.
Innate Pharma uses antibodies to enhance the immune system’s ability to fight cancer. It has formed a clinical trial collaboration with the MedImmune, an R&D arm of AstraZeneca. The two companies will share the costs of a Phase I/II study to test a combination of durvalumab with Innate’s anti-C5aR monoclonal antibody, IPH5401, for the treatment of solid tumors. Innate’s market cap of €335M will help it to support a Phase I trial to select a dose that can be used at Phase II to test the combination’s safety and efficacy.
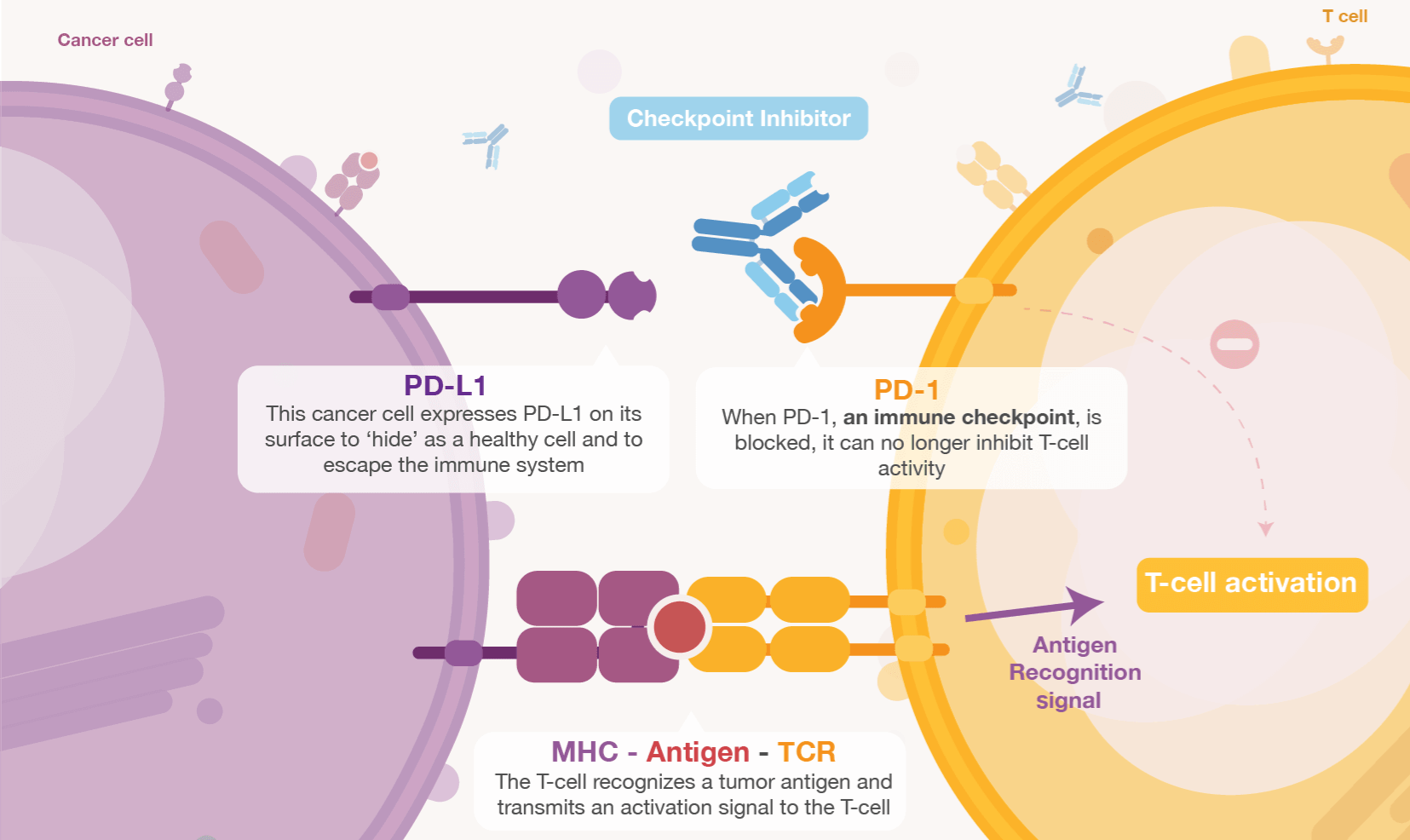
Durvalumab is a checkpoint inhibitor that blocks PD-L1 from interacting with PD-1 and CD80 to overcome the tumor’s capacity to escape the immune system. Meanwhile, Innate’s cancer antibody IPH5401 reduces the activity of complement factor 5a (C5a) – also targeted by InflaRx for the treatment of inflammatory diseases – by blocking its receptor. The C5a receptor is often overexpressed on the surface of myeloid-derived suppressor cells (MDSCs), which are immune cells found in the tumor microenvironment.
Preclinical findings indicate that blocking the C5a receptor increases immune-mediated tumor killing and the efficacy of checkpoint inhibitors. Normally, tumor cells secrete C5a, which attracts and stimulates C5a receptor-overexpressing MDSCs and neutrophils in the tumor microenvironment. These cells boost tumor growth by secreting angiogenic and immunosuppressive factors, suppressing the activity of T and NK cells, and interfering with the activity of PD-1/PD-L1 checkpoint inhibitors.
Innate’s new CEO, Mondher Mahjoubi arrived from AstraZeneca in December 2016 and it could be said that he is playing it safe by sticking to what he knows. With question marks over the efficacy of checkpoint inhibitors as a monotherapy, Innate Pharma and MedImmune may have found a way to improve their cancer-fighting capacity. In this early-stage collaboration, Innate will conduct the Phase I/II study to see if their interesting preclinical findings can translate into an effective treatment for solid tumors.
Using a combination of drugs to fight cancer is a popular approach as it could help to overcome cancer’s capacity to develop resistance to therapeutic approaches. One of our biotech dinosaurs, Transgene, received €14.4M from European and US investors to support the development of its immunotherapies in combination with checkpoint inhibitors. BerGenBio is testing its AXL kinase inhibitor in combination with the cancer antibody Keytruda, and it has achieved encouraging results in four different Phase II studies. Meanwhile, Pierre Fabre and Array BioPharma’s triple combination therapy was able to boost progression-free survival in colorectal cancer patients during a Phase III trial.
Immune checkpoint inhibitors and T-cell receptors may be a ‘match made in heaven’ for the treatment of blood cancers but researchers at Innate Pharma and MedImmune have identified a specific biological pathway that the combination of durvalumab and IPH5401 could exploit to effectively treat for solid tumors.
Images – deepadesigns / shutterstock.com
Oncology R&D trends and breakthrough innovations