Newsletter Signup - Under Article / In Page
"*" indicates required fields
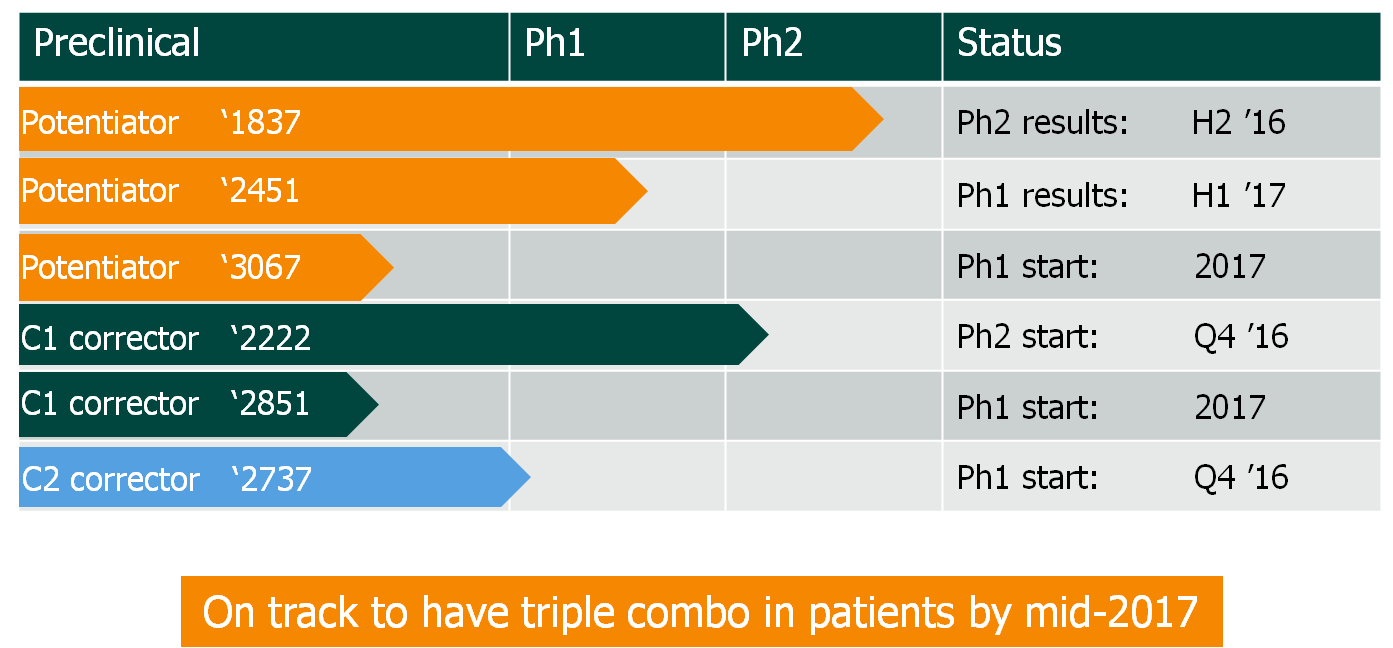
Galapagos has announced to kick off a Phase I trial with its novel Cystic Fibrosis potentiator GLPG3067, triggering a €7M milestone payment from AbbVie.
Galapagos and AbbVie teamed up in 2013, with the aim of developing a triple combination therapy for the treatment of Cystic Fibrosis (CF). Now, Galapagos announced the start of a Phase I trial with its new CF potentiator GLPG3067 both as monotherapy as well as in combination with the CF corrector GLPG2222.
Cystic Fibrosis is a life-threatening disease caused by mutations in the CFTR membrane channel that result in abnormal transport of chloride across membranes. Galapagos and AbbVie want to combine drugs with different mechanisms of action to restore the malfunction of the CFTR protein using a triple combination therapy.
The company is currently testing the combination of potentiator GLPG2451 and corrector GLPG2222 in healthy volunteers and is planning to add corrector GLPG2737 to make a triple combination. The new CF potentiator GLPG3067 serves as an addition to Galapagos’ current combination therapy portfolio.
The two companies believe that with triple combination therapies they can reach about 90% of CF patients, compared to the 50% efficacy of the current combination therapy Orkambi. Orkambi is marketed by Vertex pharmaceuticals and is a combination of a CF potentiator and a CF corrector, which is approved for patients that carry the most common CF mutation (F508del). First preclinical in vitro studies with triple combinations have shown an increased chloride transport compared to Orkambi.
If Galapagos succeeds, it may receive up to €520M ($560M) in additonal total milestone payments from AbbVie. However, the competition is currently one step ahead. Vertex is making progress with its own next-generation CFTR correctors to be used with their already approved therapy Kalydeco. Vertex has already launched two Phase II trials assessing its corrector drugs VX-440 and VX-152 in triple combination therapies in late 2016.
We continue to explore additional molecules to enrich our growing portfolio of cystic fibrosis drug candidates,” said Dr Piet Wigerinck, CSO of Galapagos. “We plan to initiate multiple studies within our CF portfolio in the course of this year, as we get closer to our goal of initiating a patient evaluation of a triple combination therapy by mid-2017.”
Images via shutterstock.com / Rene Holtslag and glpg.com






