Newsletter Signup - Under Article / In Page
"*" indicates required fields
DNAlytics changes the diagnosing game. The company builds in vitro diagnosis tests that save many years for patients! I met with Thibault, the founder, to know more about the company.
Based in Louvain-La-Neuve, Belgium, DNAlytics develops revolutionary disease diagnostic processes that complement conventional methods. Their main product is RheumaKit, a test that analyzes biological and clinical data to identify and treat arthritis disease.
Thibault Helleputte is the young founder. He started his company straight after his thesis on machine learning focused on biological data analysis. Then Thibault quickly pivoted to build innovations that could impact patient’s lives and have a larger global impact. Discover his story here:
How does RheumaKit work?
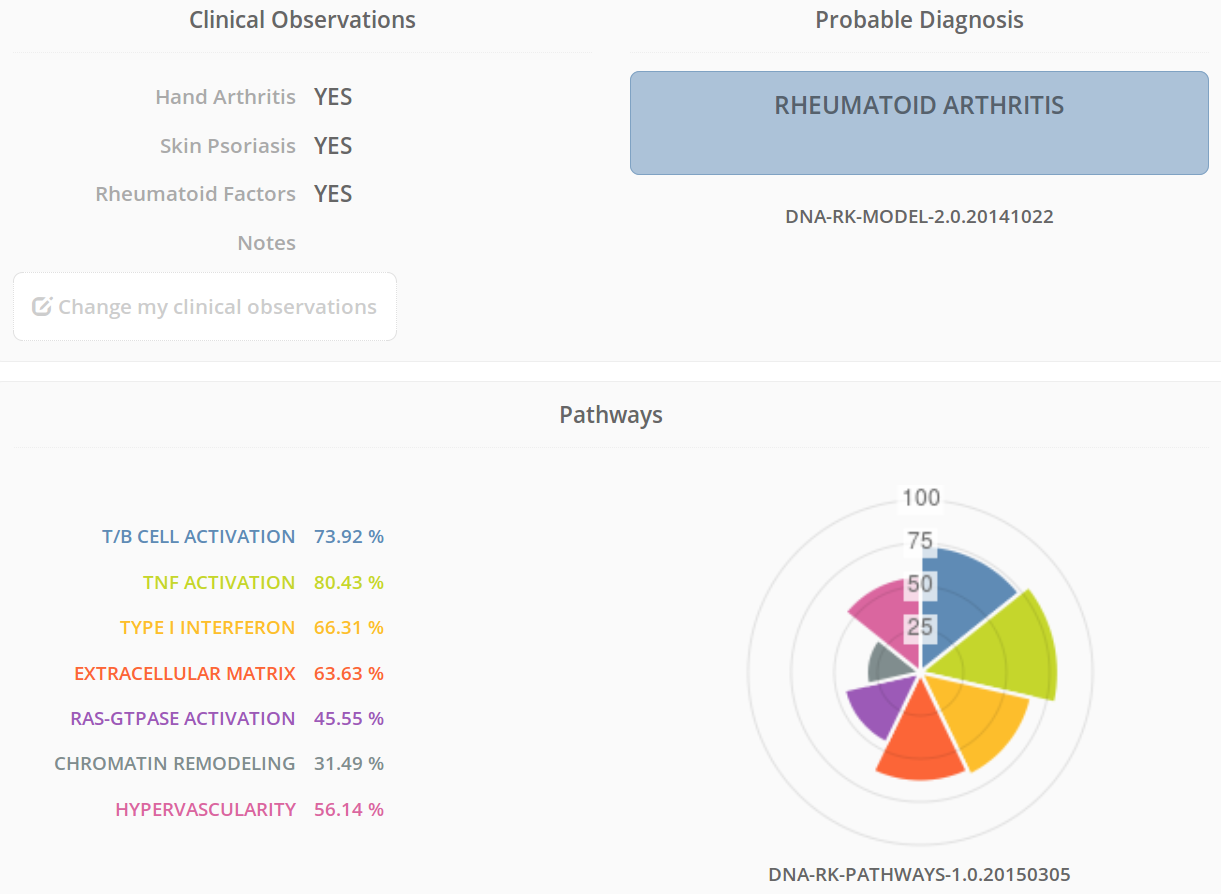
RheumaKit allows a doctor to identify exactly what kind of arthritis a patient has. Then it goes further by suggesting the most appropriate treatments mechanisms.
Here is how it works: a biopsy is harvested from joint tissue, and the sample is sent to our analytic lab. An RNA analysis is done by qPCR. In addition, the doctor has to answer a series of binary questions that add more information. We cross-reference all these data, and 15 days later, the doctor receives the complete report. The doctor also gets the picture of the metabolic pathways, shedding light on which treatment might be the best match, case by case.

How does this new generation of diagnosis help patients?
There are many forms of arthritis. In about 20% of the cases, when someone is at an early stage, it is very hard to identify precisely which kind it is. Once the right type is identified, the appropriate treatment must be chosen. For rheumatoid arthritis, a first (synthetic) treatment is applied. This only works in about 60% of the cases. For the patients who are not responding to this treatment, 2nd line drugs have to be prescribed. These are biological treatments — Disease-Modifying Anti-Rheumatic Drugs, or DMARDs.
There is a dozen of such very expensive treatments, with different mechanisms of action and all with about 60% efficacy. As there is no way for the rheumatologist to objectively choose between these treatments, a trial-and-error mechanism is often initiated. The patient loses years, and the governments pay thousands of euros for nothing. This could be avoided with our solution.
DNAlytics is developing other products, other specific diagnostic tests like a colon cancer detection test from a blood sample. How many people would prefer to give a drop of blood instead of undergoing a colonoscopy?
You started working on your thesis, then you found yourself an entrepreneur starting your own company. Tell me, how did you get to start working on DNAlytics?
During my thesis, I was developing new computer analysis methods for biological data. At its beginning, DNAlytics was a university spin-off that commercialized our expertise in biological data analysis as a consulting service. But after some time we realized that the impact we had on the world was kind of limited… Further, this consulting model is not really scalable for a startup.
So, in 2014, we decided to pivot. We moved from consulting to developing innovative products that make a real impact. This is when we decided to develop new in vitro diagnostic tests, and in 2015, we built RheumaKit.
What is your vision on personalized medicine? How do you think it could evolve in the next years?
There are two mains problems that limit personalized medicine’s evolution in Europe. First, healthcare systems should reform the medical reimbursement system. Today everything is focused on treatment, and especially on drugs, while the procedure to get a medical device or diagnostic test reimbursed is much more complicated.
It is opaque, long, and mainly only accessible to big companies. Although those non-drug solutions save a lot of money for the government and often save many years for the patient, the procedure should be way easier for SMEs and startups!
The other point is to have access to medical data. The data from health care systems are rarely available to small enterprises and to startups, if available at all. I think that the governments should be able to work hand-in-hand with all the companies to make things move faster.

As more tools become available and more data is gathered, I think personalized precision built on Machine Learning will be available. But I think that for a long time doctors will bridge the gap between machine predictions and taking the final decision. In a first stage, precision medicine will help choose the most appropriate treatment; then a custom-designed cure will be made. We already see it with cellular therapy, for instance.
Finally, we have to keep in mind more ethical questioning. Should we always understand the machine’s reasoning? What if a machine takes a decision that we do not understand? I personally think that we can get through this barrier, if these innovations still go through randomized clinical trials, just as any other medical innovation.
 Célestin de Wergifosse is a 21-year old entrepreneur and bioengineering student. As co-founder of Young Change Maker, his dream is to change the world with biotechnology products by discovering biotech advances in untapped areas like Central and South Asia.
Célestin de Wergifosse is a 21-year old entrepreneur and bioengineering student. As co-founder of Young Change Maker, his dream is to change the world with biotechnology products by discovering biotech advances in untapped areas like Central and South Asia.
Images from DNAnalytics / author & Bruce Rolff, crystal light, ImageFlow, Khakimullin Aleksandr / shutterstock.com






