Newsletter Signup - Under Article / In Page
"*" indicates required fields
By this point, you can’t have escaped hearing about CRISPR. First discovered in the 1980’s, the repeated sequences and their function in bacterial defense systems remained a mystery until almost 10 years ago, when it was hailed as the biggest scientific discovery of the century. Since then, scientists have leveraged the adaptable, heritable mechanism to develop the most powerful gene-editing technology to date, known as CRISPR-Cas9. But the implementation of CRISPR may hinge on a crucial technology, the lentiviral vector!
In collaboration with
How do the ‘DNA Scissors’ Work?
There are three major parts to this system: the cutting enzyme, Cas9; guide RNA, to guide Cas9; and the DNA to be cut. The guide RNA is able to direct Cas9 by having a specific sequence, in this case the repeat that gives CRISPR its name, which complements a portion of the target DNA.
These strands combine, and Cas9 goes to work, cutting both strands of the DNA, implanting new DNA, and stitching it back together. Cas9 can also be modified with the addition of a protein to activate or repress specific genes. This is a highly specific tunable mechanism, as the enzyme targets specific motifs on either side of the viral DNA using own strand of RNA (crRNA). Scientists have since developed it into a revolutionary technology to edit the human genome.
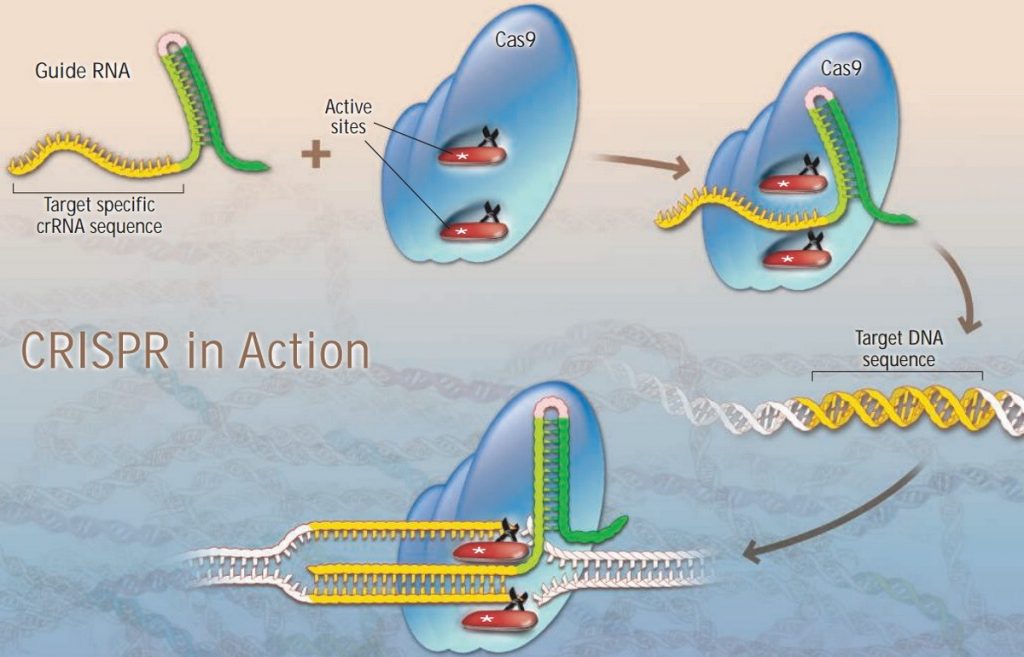
Up until this point, the only competing editing technologies were zinc fingers and TALEN enzymes, both of which require the labour-intensive generation of entire proteins. (That said, TALEN has since been developed into a more precise tool that has made it to clinical trials.)
While improvements need to be made in its specificity, CRISPR-Cas9 is easier to manipulate as an RNA-based technique and therefore much simpler and faster than either of its predecessors. This improvement is crucial for high throughput gene editing tests to adapt treatments quickly to fit the needs of a patient. But it’s not much use if it’s impossible to deliver the technology to therapeutic targets!
For all of its promise, the application of CRISPR remains limited because of inefficient delivery. This step is crucial: the technology isn’t worth much if it’s impossible to deliver the technology to therapeutic targets!
So how do we make CRISPR useful?
There are two scenarios that pose problems for CRISPR, and unfortunately, they are both crucial in therapeutic development. The first is in vitro applications to certain refractory cells like primary cells or Hematopoietic stem cells; the second is in vivo applications in general. One possible solution to increase the efficiency and make a CRISPR-based therapy viable is to incorporate it into a viral vector, like a lentivirus.
A lentivirus belongs to a subclass of retroviruses characterized by an envelope, a capsid and a RNA genome inside of these. This virus targets specific cells or tissues with a very high efficiency and selectivity. Once inside the cell, RNA genome is reverse-transcribed in DNA and integrated into the genome of the cell, thanks to the viral integrase enzyme.
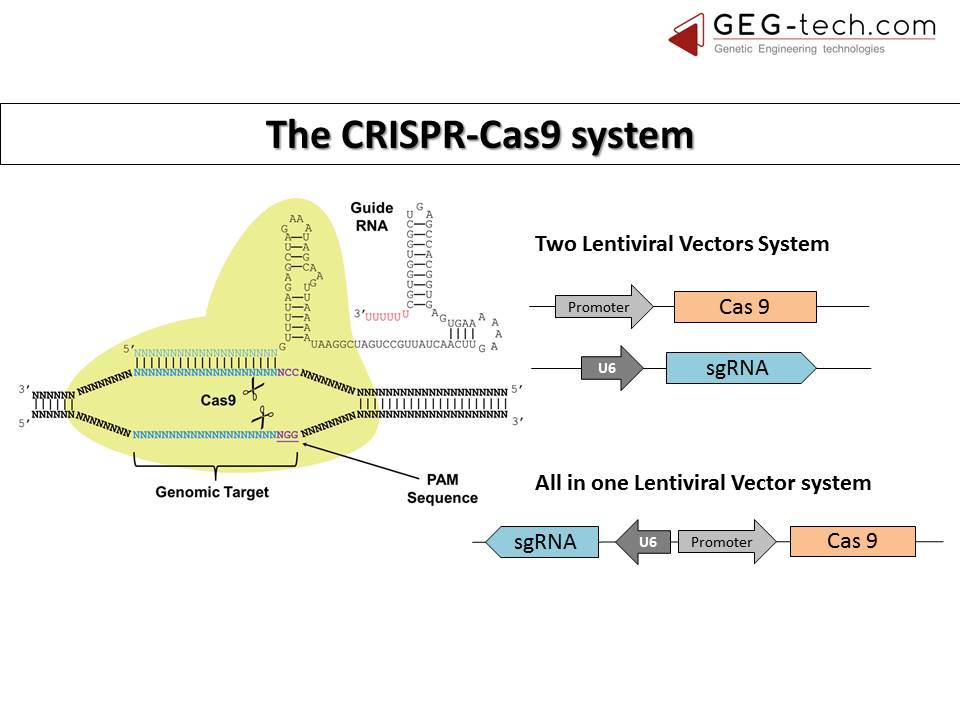
Given that such an efficient gene transfer tool already exists in nature, it seems an almost obvious choice to use it in tandem with CRISPR! These lentiviral vectors are extra efficient because of their specificity in targeting a certain cell or tissue and large cargo capacities.
There are two versions of this system. The first is an integrating lentiviral vector, which induces long and robust expression and is useful for screening or validating experimental parameters; the other is its non-integrating counterpart that induces transient expression and is used mostly for applications. Both offer very high delivery efficiency, and the integrating system stands out as an “all-in-one” technology by combining both cas9 and the guide RNA.
Where can I get these lentiviral vectors?
Transforming a virus into a safe and efficient vector is a long process requiring lots of specialist expertise. Finding a quality vector that will work in your experiment can be tricky, so choosing a good vector provider is an important step that should be carried out with due diligence.
Fortunately, we can recommend GEG-Tech as a reliable provider. The French company has developed Lenti-ONE products, a collection of lentiviral vectors that can be developed to express the CRISPR system. With extensive experience under its belt, GEG Tech allows it to make a real lentivirus toolkit that fits its customer’s needs.
The GEG-Scientists may be the ‘Geeks of the Lentiviral Vector Technology’, as they take pride in fine-tuning and adapting its features to generate different tools. For example, they played with the virus’ envelope to broaden or restrict the range of targeted cells; in another, they mutated the viral integrase to obtain different mutants which are non-integrating. As a third avenue of development, the team blocked the reverse transcription step to obtain a lentiviral vector entirely RNA, and this became the foundation of GEG Tech’s latest platform, Lenti-One Trans.

These examples demonstrate the benefits of the lentiviral vectors: efficiency, specificity, high cargo load, and low immunogenicity. Expression without DNA phase, as in these cases, confer a high level of biosafety upon the technology, that together with the other advantages, makes lentiviral vectors a powerful complement to CRISPR.
For a better understanding of the lentivirus toolkit and the different processes opened up by it, GEG Tech shares comprehensive videos, knowledge, and protocols on their website. Customers can also directly choose a vector of interest from their broad online catalogue of over 4000 items or customize a new one on the company’s online platform. And help is never far from reach – a customer can always check with GEG Tech experts before ordering about vector design and project feasibility.
If you’d like more information, visit their website or meet GEG-Tech through their social networks.
Hopefully, we alerted you to the current issues in turning CRISPR into a viable therapy! Since the gene editing technology’s development has been called the ‘scientific discovery of the century’, we’re sure that CRISPR will hit it big in biotech. Stay tuned for news on further developments as it makes its way through the clinic!
Reference, Figure 1: The CRISPR Craze, Elizabeth Pennisi, 2013. Science 341 no. 6148 pp. 833-836
Featured Image, Figure 2 right side: GEG Tech
Figure 2 left side: James E DiCarlo. Nucleic Acids Research, 2014, Vol. 42, No. 19. doi: 10.1093/nar/gku749
Figure 3: Mopic/shutterstock.com







