Newsletter Signup - Under Article / In Page
"*" indicates required fields
People with type 1 diabetes need lifelong treatments of daily insulin injections to manage their condition that still leave them at risk of long-term complications. Immunotherapy could one day become an insulin-free alternative to stop, prevent, and potentially cure this chronic disease.
Type 1 diabetes is an autoimmune disease where the insulin-producing beta cells in the pancreas are wrongly detected as foreign and destroyed by the immune system.
“There is no cure; once initiated, the disease will progress to complete destruction of the insulin-producing cells of the pancreas,” Pierre Vandepapelière, previous CEO of Imcyse, told me. This Belgian company is developing an innovative form of treatment that could change the way type 1 diabetes is treated.
Currently, the standard treatment for the disease consists of monitoring glucose levels and frequent insulin injections to keep healthy blood sugar levels. However, even with the best control measures, patients are still at risk of complications affecting the eyes, kidneys and nerves in the long term. Insulin treatment also carries the risk of inducing episodes of extremely low blood sugar, also known as hypoglycemia, which can be life-threatening.
Searching for alternatives
There is currently research focusing on automating the process of measuring glucose and injecting the right amount of insulin through a so-called ‘artificial pancreas’. That solution is still not ideal, though. “You would still be dependent on insulin and glucose measurement, and even the best closed loops would come with a risk of hypoglycemia,” said Jacob Sten Petersen, Corporate Vice President and Head of Stem Cell R&D at Novo Nordisk.
As a leader in diabetes treatments, Novo Nordisk is researching technology with potential to cure type 1 diabetes, eliminating insulin injections and long-term complications. The company is studying the transformation of stem cells into insulin-producing cells that can be transplanted into the pancreas to recover normal insulin production.
“Just like endogenous beta cells, implanted, stem cell-derived beta cells have the ability to sense changes in blood glucose levels in real time, and to secrete appropriate amounts of insulin until glucose levels are normalized,” said Cord Dohrmann, CSO of Evotec. This German biotech company is working with Sanofi to develop a cell-replacement therapy for type 1 diabetes.
Beta-cell replacement has been studied for a long time, and was originally carried out using insulin-producing cells from donors. However, this treatment faces a big challenge. The immune system that once rejected the original beta cells will also reject the transplanted cells. This means that patients need to go on lifelong immunosuppression. Some are researching ways of encapsulating the cells to protect them, as is the case of companies like the French Defymed or US-based Viacyte, but these approaches are still at a very early stage. However, in the last few years a new alternative has started gaining attention.
Immunotherapy: attacking the origin
Immunotherapy is mostly known as a modern treatment for allergies, where a person is exposed to a small amount of what they are allergic to in order to train the immune system to tolerate it. The principle of modulating the immune system to either attack or tolerate specific targets has gained a lot of recognition in recent years thanks to its potential to treat all sorts of diseases where the immune system is involved, including cancer and neurodegenerative conditions.
As autoimmune diseases are directly caused by the immune system, immunotherapies have started being investigated as an approach to treat a number of different autoimmune conditions, among them type 1 diabetes.
Killing rebel immune cells
“In type 1 diabetes, cells in the immune system wrongly recognize the insulin-producing cells in the pancreas as foreign and dangerous,” said Vandepapelière. “Our objective is to interrupt this autoimmune process.”
Imcyse is developing an immunotherapy that involves injecting a molecule that makes the system produce a new type of immune cell. These cells will specifically kill those immune cells that destroy the pancreas.
“The mechanism is specific to type 1 diabetes and to the pancreas, therefore not affecting the general immune defenses, as would classic immunosuppressive drugs, or other organs,” added Vandepapelière.
If effective, the treatment could stop the destruction of insulin-producing cells. The first to benefit would be people recently diagnosed with the disease. “Early after diagnosis, between 3 to 6 months, it is estimated that around 10% of the insulin-producing cells are still alive and producing insulin. After stopping the autoimmune process, the remaining beta cells would be protected and could continue producing insulin,” said Vandepapelière.
There is also potential that this could in turn promote the natural regeneration of the remaining beta cells, but that has to be confirmed in clinical trials. Imcyse is currently running a phase Ib trial that is recruiting patients that were diagnosed with type 1 diabetes less than 6 months before. Early results suggest the therapy shows promise and it should move into phase II testing this year.
Vaccinating against type 1 diabetes
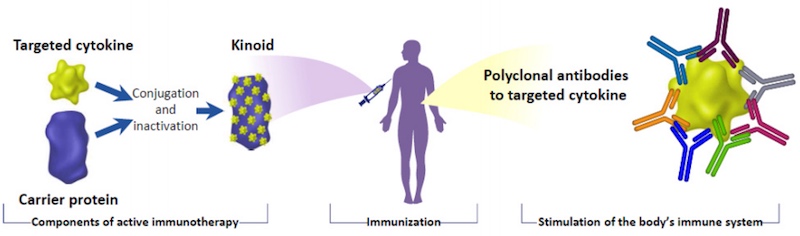
“Autoimmune diseases are characterized very often by an overexpression of a specific cytokine; a specific protein that we do produce normally, but which in case of disruption of the immune system is produced in excess,” Miguel Sieler, previous CEO of Neovacs, told me.
Based in France, Neovacs aims to block this excess cytokine production to stop autoimmune diseases in their tracks. To do so, the company is developing a vaccine that stimulates the immune system to neutralize the specific protein that is being produced in excess.
In the case of type 1 diabetes, that protein is known as interferon alpha. In excess, it induces the appearance of immune cells that attack and destroy insulin-producing cells. “We can stop type 1 diabetes by neutralizing the excess of interferon alpha,” said Sieler. “It’s the same principle as a viral vaccine.”
Neovacs has already tested the same vaccine in people with lupus, another autoimmune disease. Results have shown that the vaccine can keep the patients immunized for 5 years. This means that a person with type 1 diabetes would only need to receive an initial treatment of 6 months and then a booster every 4 to 5 years.
“This would make treatment with insulin no longer necessary, and of course reduces the cost considerably compared to a lifetime treatment,” explained Sieler.
Teaming up with bacteria
The bacteria in our gut have a strong connection with our immune system. ActoBio, a company based in Belgium, wants to exploit this connection to treat type 1 diabetes. “Our product candidates are based on the use of Lactococcus lactis bacteria that are genetically modified to express proteins, peptides, antibodies or human cytokines,” Lothar Steidler, CTO of ActoBio, told me.
Our guts are familiar to this bacterium, as it’s used to produce cheese and buttermilk. In the case of type 1 diabetes, the bacteria are engineered to produce the insulin precursor human proinsulin and a signaling molecule called interleukin 10. Together they signal the gut-associated lymphoid tissue, where immune cells are stored, to restore the tolerance of insulin-producing cells with the goal of slowing down or stopping their destruction.
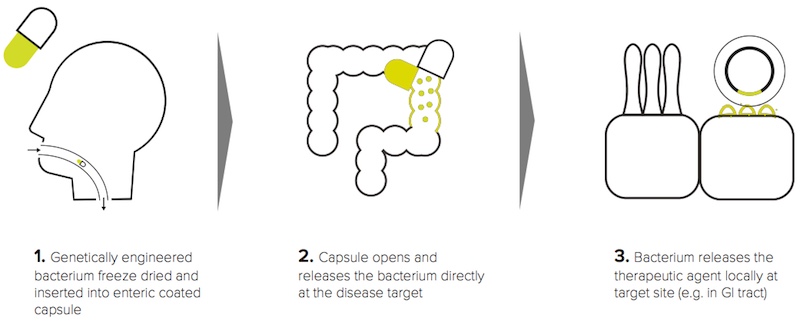
“It is potentially a safe oral treatment that will be given for a limited period of time and could lead to patients who develop type 1 diabetes not needing to use insulin, or delay the need for insulin after diagnosis” added Pieter Rottiers, CEO of ActoBio.
The company is now running a phase I/II clinical trial in the US and Europe in people with type 1 diabetes as young as 12 years old. “Patients will have to have some remaining capacity for production of insulin,” said Rottiers. Results are expected this year.
Towards the future: prevention and cure
Although the first goal of immunotherapy treatments for type 1 diabetes is to stop the progression of the disease in people who still have some insulin-producing cells, this technology has potential to go beyond that.
Immunotherapy could also prevent the development of type 1 diabetes in people known to be at risk. “The risk of developing type 1 diabetes can be predicted 3 to 5 years before its onset,” said Vandepapelière. “It could therefore be possible to prevent the disease by halting this autoimmune process early. If conducted on a nationwide scale, this could possibly eradicate the disease.”
Imcyse is contemplating testing the ability of its immunotherapy to prevent type 1 diabetes. To do so, it would have to implement a wide screening, particularly in children, to identify the subjects most at risk of developing the disease. This, however, would take considerable time. “A longer study is needed to demonstrate the preventive efficacy, as with most vaccine developments,” said Vandepapelière.
Further down the line, but already a tangible possibility, immunotherapy could be the key to the much-wanted cure for type 1 diabetes. “In patients with established type 1 diabetes, an extinction of the autoimmune and inflammatory process could regenerate the beta cells, either spontaneously or after grafting beta cells,” said Vandepapelière.
It still has to be demonstrated that immunotherapy can actually be a cure for type 1 diabetes. It will likely take years. Still, one thing is sure; as Vandepapelière put it: “In the future, treatment of type 1 diabetes will not remain limited to controlling the disease by administration of insulin.”
This article was originally published in January 2019 and has since been updated to reflect the latest developments.
Images via Shutterstock, Neovacs, and ActoBio






