Newsletter Signup - Under Article / In Page
"*" indicates required fields
Gene therapy is a hot area in the biotech industry right now, with many treatments in development and a number of recent approvals. However, the path has not always been a smooth one.
Gene therapy has been one of the biggest success stories of the 21st century. Genetic diseases were once seen as incurable, etched in stone (or rather, in nucleotides) within the genomes of those unfortunate enough to receive them in life’s genetic lottery. Not anymore.
Over the last few years, there has been a near constant stream of positive news around gene therapy, with treatment after treatment receiving regulatory approval, succeeding in trials, or raising vast sums to enter development. And with over 1,000 clinical trials currently underway, we may finally be witnessing the long-awaited arrival of the gene therapy revolution.
The field has had a long and often bumpy road to get to this point, with both triumph and tragedy along the way. Let’s take a look back at gene therapy’s evolution from futuristic idea to tangible treatment, and what is still to come.
The early years
In 1972, a paper titled ‘Gene therapy for human genetic disease?’ was published in Science by US scientists Theodore Friedmann and Richard Roblin, who outlined the immense potential of incorporating DNA sequences into patients’ cells for treating people with genetic disorders. However, they urged caution in the development of the technology, pointing out several key bottlenecks in scientific understanding that still needed to be addressed.
“For the foreseeable future… we oppose any further attempts at gene therapy in human patients because (i) our understanding of such basic processes as gene regulation and genetic recombination in human cells is inadequate; (ii) our understanding of the details of the relation between the molecular defect and the disease state is rudimentary for essentially all genetic diseases; and (iii) we have no information on the short-range and long-term side effects of gene therapy.”
Following 18 years of further research, the first gene therapy trial launched in 1990. A four-year-old girl named Ashanthi DeSilva underwent a 12-day treatment for a rare genetic disease known as severe combined immunodeficiency. DeSilva lacked a key enzyme called adenosine deaminase (ADA), which left her immune system crippled and put her at constant risk of contracting an infection that could kill her.
A viral vector was used to introduce a functional copy of the gene encoding the ADA enzyme into DeSilva’s immune cells. This improved her immune system’s function and allowed her to live a normal life, without having to be isolated to avoid infection.
The success of DeSilva’s case was a major milestone, and numerous further trials were enthusiastically launched throughout the 1990s. The atmosphere of optimism didn’t last though. Nine years later gene therapy encountered a devastating setback — the first reported death of a patient during a clinical trial.

In 1999, 18-year-old Jesse Gelsinger had signed up for an experimental gene therapy trial at the University of Pennsylvania. He had a genetic condition known as ornithine transcarbamylase deficiency. The disease, caused by a genetic mutation, compromised his liver’s ability to break down toxic ammonia, which accumulated in his blood.
The trial was designed to introduce a working copy of the missing gene into his liver cells using an adenovirus (a modified common cold virus) to deliver it. Four days after being treated, Gelsinger died, having suffered a catastrophic immune reaction to the treatment.
Gelsinger’s death shocked the entire field and drew significant media attention. The US FDA criticized the design of the trial, suspended the university’s entire gene therapy program (one of the largest in the world at the time), and launched investigations into 69 other gene therapy trials taking place across the country. The safety of viral vectors came under high levels of scrutiny. Gene therapy was seen to have moved too far too fast. A slower, more cautious approach was urged.
When the field finally rebounded, it did so gradually. China became the first nation to approve a gene therapy, called Gendicine, for head and neck cancer in 2003. This was followed by Russia’s approval of Neovasculgen for peripheral artery disease in 2011. In 2012, the European Commission granted approval to uniQure’s Glybera for treating the ultra-rare disease lipoprotein lipase deficiency.
While initially celebrated as a breakthrough moment for gene therapy in the EU, Glybera was largely a commercial failure. With a price of €1M, it was the most expensive treatment in the world. In 2017, the therapy was withdrawn after being prescribed to a single patient.
From bust to boom
Since the first wave of gene therapy approvals between 2003 and 2012, the pace has rapidly picked up. Half a dozen gene therapies have arrived in the EU over the last few years, most recently Zynteglo for the blood disorder beta-thalassemia. The US has experienced a similar boom, and the FDA expects an approval rate of 10 to 20 cell and gene therapies every year by 2025.
Much of gene therapy’s recent success can be attributed to considerable advances in viral vector technologies used to deliver the genetic material.
“Following the early failure of gene therapy trials with retroviral vectors and highly immunogenic adenovirus vectors in the late 90s and early 2000s, huge gaps have been closed concerning virus biology, vector dynamics, immune interaction, and vector safety,” said Christian Thirion, founder and CTO of the German viral vector developer Sirion Biotech.
In particular, a class of viral vectors called adeno-associated viruses (AAVs) has emerged as a leading platform for developing gene therapies.
“AAVs are the new superstars in the gene therapy sector,” said Thirion. “Wildtype AAVs don’t elicit disease in humans and result in long-term gene expression for up to 10 years. They can be engineered and targeted towards specific cell or tissue types. All these features make them ideal tools for modern gene therapy applications, and the rise in interest in this technology has been steep.”
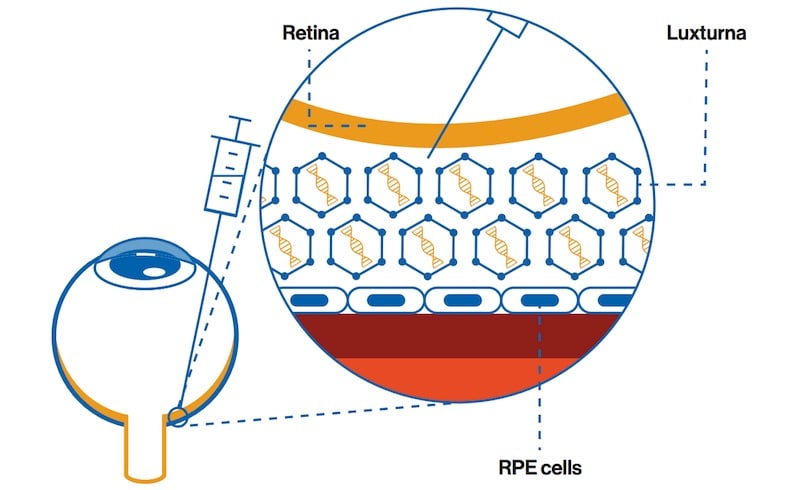
Targeting the eye has become particularly attractive, as its anatomy is well suited to gene therapy approaches. “The eye is a closed system; once injected in the eyeball, the vectors have very little opportunity to leak outside and as a result vectors will stay within the eye and be able to unload their DNA cargo into a maximum number of targeted cells. Furthermore, neurons are not renewed and once a neuron is expressing the gene of interest it is likely to do so for a very long period of time,” said Bernard Gilly, CEO of Paris-based Gensight Biologics, a company that specializes in gene therapy for retinal and neurodegenerative diseases.
The 2018 approval of Luxturna in the EU was the first for any gene therapy for the eye. The treatment consists of an AAV-based gene therapy to stop the progression of vision loss caused by a genetic mutation.
“Luxturna’s approval was a significant milestone for the industry, our company, and others who are looking to offer new therapies in ophthalmology,” said Gilly. In Europe, companies such as Nightstar Therapeutics, Horama, Eyevensys, and MeiraGTx are developing multiple gene therapies targeting different genetic mutations that cause blindness.
A CRISPR future?
The idea of simply ‘editing out’ genetic diseases has been one of the most widely discussed applications of the revolutionary CRISPR gene editing technology. This technique has immense potential and could broaden the concept of genetic treatment beyond conventional gene therapy approaches.
A number of key players have emerged in the race to develop the first CRISPR-based therapy, including Editas Medicine, Intellia Therapeutics, and CRISPR Therapeutics.
“Many traditional gene therapies introduce a functional copy of a gene into a cell, but do not reduce or stop expression of the original diseased gene,” a spokesperson for CRISPR Therapeutics told me. “Gene editing approaches can alter the DNA to precisely disrupt, delete, or repair the original diseased gene. As a result, gene editing has the potential to address a vast array of genetic disorders.”
In partnership with Vertex Pharmaceuticals, CRISPR Therapeutics launched the first CRISPR trial in humans for the treatment of beta-thalassemia and sickle cell disease in 2018. As of last year, preliminary results seem to indicate the treatment works. The company is also developing several CRISPR therapies for cancer and regenerative medicine.
“There has been a rapid technology cycle to make this platform a reality and to allow us to begin bringing the first CRISPR-based medicines to patients. We have laid a careful groundwork with our preclinical research and are very optimistic about what the next phase means for science and for patients.”
Gene therapy has overcome immense challenges to become a medical reality, and its evolution is still far from over. Increasingly powerful and elegant molecular tools will continue to expand our ability to correct genetic disorders, offering fresh hope to patients around the world.
Hurdles still remain, especially around the question of how to fairly price these costly treatments, but it’s clear that after nearly 50 years of effort, gene therapy’s transformative potential is finally being realized.
This article was originally published in June 2019 and has since been updated to reflect the latest advancements in the field of gene therapy. Cover illustration by Elena Resko, images from Shutterstock and Novartis.






